Международный эндокринологический журнал Том 19, №4, 2023
Вернуться к номеру
Поліморфізм TaqI гена рецептора вітаміну D у дітей із соматотропною недостатністю
Авторы: O.V. Bolshova (1), M.O. Ryznychuk (2), D.A. Kvachenyuk (1)
(1) — State Institution “V.P. Komisarenko Institute of Endocrinology and Metabolism of the National Academy of Medical Sciences of Ukraine”, Kyiv, Ukraine
(2) — Bukovinian State Medical University, Chernivtsi, Ukraine
Рубрики: Эндокринология
Разделы: Клинические исследования
Версия для печати
Актуальність. Ядерний рецептор вітаміну D опосередковує більшість біологічних функцій цього вітаміну. Він належить до сімейства рецепторів стероїдних гормонів, ген якого (VDR) розташований на хромосомі 12q13.1. Повідомлялося про генетичну мінливість гена VDR, у якому ідентифіковано понад 470 однонуклеотидних поліморфізмів. Одним з найпоширеніших поліморфізмів у гені VDR є rs731236 (TaqI). Мета дослідження: вивчити поліморфізм TaqI гена VDR у дітей із недостатністю гормону росту (НГР). Матеріали та методи. Визначення TaqI поліморфізму гена VDR (rs731236) проводили за допомогою методу полімеразної ланцюгової реакції з наступним аналізом довжини рестрикційних фрагментів при виявленні їх шляхом електрофорезу в агарозному гелі у 28 дітей препубертатного віку із НГР. Результати. У групі хворих із НГР частка гетерозигот Т/С TaqI поліморфізму гена VDR (rs731236) в 1,28 раза вища, ніж серед здорових осіб. Пацієнтів — носіїв генотипу Т/Т та С/С у 0,68 та 0,90 раза менше, ніж у контрольній групі. Наявність гомозиготного генотипу ТТ підвищує ризик розвитку НГР, але невірогідно (відношення шансів (ВШ) = 1,89; 95% довірчий інтервал (ДІ) 0,66–5,39; р = 0,23), а наявність гомозиготного генотипу СС є протективною (ВШ = 0,75; 95% ДІ 0,17–3,22; р = 0,70). При аналізі алелей у пацієнтів із НГР отримані наступні дані: носійство алеля Т поліморфного локусу rs731236 TaqI гена рецептора вітаміну D асоціюється з ризиком соматотропної недостатності (ВШ = 1,24; 95% ДІ 0,65–2,36; р = 0,52), але не досягає статистичної вірогідності. Співвідношення частот алелів (pТ = 0,554, qС = 0,446) практично не відрізняється від 1 : 1, що свідчить про збереження частоти алелів в українській популяції. Висновки. У дітей із НГР частка генотипу Т/С в 1,28 раза вища, ніж у здорових осіб. Наявність гомозиготного генотипу ТТ підвищує ризик розвитку НГР, але невірогідно (ВШ = 1,89; 95% ДІ 0,66–5,39; р = 0,23). Носійство алеля Т поліморфного локусу rs731236 TaqI гена VDR асоціюється з ризиком соматотропної недостатності (ВШ = 1,24; 95% ДІ 0,65–2,36; р = 0,52).
Background. The nuclear receptor for vitamin D mediates most of the biological functions of this vitamin. It belongs to the steroid hormone receptor family, the gene for which (vitamin D receptor — VDR) is located on chromosome 12q13.1. Genetic variability has been reported in the VDR gene, in which more than 470 single nucleotide polymorphisms have been identified. One of the most common polymorphisms in the VDR gene is rs731236 (TaqI). The purpose was to study the TaqI polymorphism of the VDR gene in children with growth hormone deficiency (GHD). Materials and methods. The TaqI polymorphism of the VDR gene (rs731236) was determined using the polymerase chain reaction, followed by analysis of the length of restriction fragments detected by agarose gel electrophoresis in 28 prepubescent children with GHD. Results. In the group of patients with GHD, the proportion of heterozygotes for T/C TaqI polymorphism of the VDR gene (rs731236) is 1.28 times higher than among healthy individuals. There were 0.68 and 0.90 times fewer patients carrying T/T and C/C genotypes than in the control group. The presence of a homozygous TT genotype increases the risk of developing GHD, but not significantly (odds ratio (OR) = 1.89, 95% confidence interval (CI) 0.66–5.39; p = 0.23), and the presence of a homozygous CC genotype is protective (OR = 0.75, 95% CI 0.17–3.22; p = 0.70). When analyzing alleles in patients with GHD, the following data were obtained: carriage of the T allele for the polymorphic loci TaqI rs731236 of the VDR gene is associated with the risk of GHD (OR = 1.24, 95% CI 0.65–2.36; p = 0.52) but not significantly. The ratio of allele (pТ = 0.554, qС = 0.446) frequencies practically does not differ from 1 : 1, which indicates the preservation of allele frequencies in the Ukrainian population. Conclusions. In children with GHD, the proportion of the T/C genotype is 1.28 times higher than in the group of healthy persons. The presence of a homozygous TT genotype increases the risk of developing GHD but not significantly (OR = 1.89, 95% CI 0.66–5.39; p = 0.23). Carriage of the T allele for the polymorphic locus TaqI rs731236 of the VDR gene is associated with the risk of the growth hormone deficiency (OR = 1.24, 95% CI 0.65–2.36; p = 0.52) but not significantly.
соматотропна недостатність; діти; TaqI поліморфізм гена рецептора вітаміну D; розподіл генотипів
growth hormone deficiency; children; TaqI polymorphism of the vitamin D receptor gene; distribution of genotypes
Introduction
Materials and methods
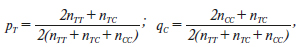
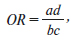
Results
Discussion
Conclusions
- Wang Y., Zhu J., DeLuca H.F. Where is the vitamin D receptor? Arch. Biochem. Biophys. 2012 Jul 1. 523(1). 123-33. doi: 10.1016/j.abb.2012.04.001.
- Tuoresmäki P., Väisänen S., Neme A., Heikkinen S., Carlberg C. Patterns of genome-wide VDR locations. PLoS One. 2014 Apr 30. 9(4). e96105. doi: 10.1371/journal.pone.0096105.
- Uitterlinden A.G., Fang Y., van Meurs J.B., van Leeu–wen H., Pols H.A. Vitamin D receptor gene polymorphisms in relation to vitamin D-related disease states. J. Steroid Biochem. Mol. Biol. 2004 May. 89–90 (1–5). 187-93. doi: 10.1016/j.jsbmb.2004.03.083.
- Pankiv V.I., Yuzvenko T.Yu., Pankiv I.V. Type 2 diabetes mellitus and subclinical hypothyroidism: focusing on the role of cholecalciferol. Problems of Endocrine Pathology. 2019. 2. 46-51. doi: 10.21856/j-PEP.2019.2.07.
- Cieślińska A., Kostyra E., Chwała B., Moszyńska-Dumara M., Fiedorowicz E. et al. Vitamin D receptor gene polymorphisms associa–ted with childhood autism. Brain Sci. 2017 Sep 9. 7(9). 115. doi: 10.3390/brainsci7090115.
- Bhanushali A.A., Lajpal N., Kulkarni S.S., Chavan S.S., Bagadi S.S., Das B.R. Frequency of FOKI and TAQI polymorphism of vitamin D receptor gene in Indian population and its association with 25-hydroxyvitamin D levels. Indian J. Hum. Genet. 2009 Sep. 15(3). 108-13. doi: 10.4103/0971-6866.60186.
- Osman E., Al Anouti F., El Ghazali G., Haq A., Mirgani R., Al Safar H. Frequency of rs731236 (Taql), rs2228570 (Fok1) of vitamin-D receptor (VDR) gene in Emirati healthy population. Meta Gene. 2015 Sep 15. 6. 49-52. doi: 10.1016/j.mgene.2015.09.001.
- Hustmyer F.G., DeLuca H.F., Peacock M. ApaI, BsmI, EcoRV and TaqI polymorphisms at the human vitamin D receptor gene locus in Caucasians, blacks and Asians. Hum. Mol. Genet. 1993 Apr. 2(4). 487. doi: 10.1093/hmg/2.4.487.
- Emmanouilidou E., Galli-Tsinopoulou A., Kyrgios I., Gbandi E., Goulas A. Common VDR polymorphisms and idiopathic short stature in children from northern Greece. Hippokratia. 2015 Jan-Mar. 19(1). 25-9.
- Steppe L., Bülow J., Tuckermann J., Ignatius A., Haffner-Luntzer M. Bone mass and osteoblast activity are sex-dependent in mice lacking the estrogen receptor α in chondrocytes and osteoblast progenitor cells. Int. J. Mol. Sci. 2022 Mar 7. 23(5). 2902. doi: 10.3390/ijms23052902.
- Fletcher D., Faddy M. Confidence Intervals for Expec–ted Abundance of Rare Species. Journal of Agricultural, Biological, and Environmental Statistics. 2007. 12(3). 315-324. Available from: http://www.jstor.org/stable/27595645.
- Stranger B.E., Stahl E.A., Raj T. Progress and promise of genome-wide association studies for human complex trait genetics. Genetics. 2011 Feb. 187(2). 367-83. doi: 10.1534/genetics.110.120907.
- Nikolac Gabaj N., Unic A., Miler M., Pavicic T., Culej J., Bolanca I. et al. In sickness and in health: pivotal role of vitamin D. Biochem. Med. (Zagreb). 2020 Jun 15. 30(2). 020501. doi: 10.11613/BM.2020.020501.
- Zhao S., Gardner K., Taylor W., Marks E., Goodson N. Vitamin D assessment in primary care: changing patterns of tes–ting. London J. Prim. Care (Abingdon). 2015. 7(2). 15-22. doi: 10.1080/17571472.2015.11493430.
- Yang A., Lv Q., Chen F., Wang D., Liu Y., Shi W. Identification of recent trends in research on vitamin D: a quantitative and co-word analysis. Med. Sci. Monit. 2019 Jan 22. 25. 643-55. doi: 10.12659/MSM.913026.
- Haussler M.R., Whitfield G.K., Kaneko I., Haussler C.A., Hsieh D. et al. Molecular mechanisms of vitamin D action. Calcif. Tissue Int. 2013 Feb. 92(2). 77-98. doi: 10.1007/s00223-012-9619-0.
- Singh I., Lavania M., Pathak V.K., Ahuja M., Turan–kar R.P. et al. VDR polymorphism, gene expression and vitamin D levels in leprosy patients from North Indian population. PLoS Negl. Trop. Dis. 2018 Nov 27. 12(11). e0006823. doi: 10.1371/journal.pntd.0006823.
- Kow M., Akam E., Singh P., Singh M., Cox N. et al. Vitamin D receptor (VDR) gene polymorphism and osteoporosis risk in White British men. Ann. Hum. Biol. 2019 Aug. 46(5). 430-3. doi: 10.1080/03014460.2019.1659851.
- Jiang L.L., Zhang C., Zhang Y., Ma F., Guan Y. Associations between polymorphisms in VDR gene and the risk of osteoporosis: a meta-analysis. Arch. Physiol. Biochem. 2022 Dec. 128(6). 1637-44. doi: 10.1080/13813455.2020.1787457.
- Haussler M.R., Whitfield G.K., Haussler C.A., Hsieh J.C., Thompson P.D. et al. The nuclear vitamin D receptor: biological and molecular regulatory properties revealed. J Bone Miner. Res. 1998. 13(3). 325-49. doi: 10.1359/jbmr.1998.13.3.325.
- Xiong D.H., Xu F.H., Liu P.Y., Shen H., Long J.R. еt al. Vitamin D receptor gene polymorphisms are linked to and associated with adult height. J. Med. Genet. 2005. 42(3). 228-34. doi: 10.1136/jmg.2004.024083.