Международный эндокринологический журнал Том 19, №4, 2023
Вернуться к номеру
Роль дефіциту холекальциферолу в розвитку латентного автоімунного діабету дорослих
Авторы: I.O. Tsaryk, N.V. Pashkovska
Bukovinian State Medical University, Chernivtsi, Ukraine
Рубрики: Эндокринология
Разделы: Клинические исследования
Версия для печати
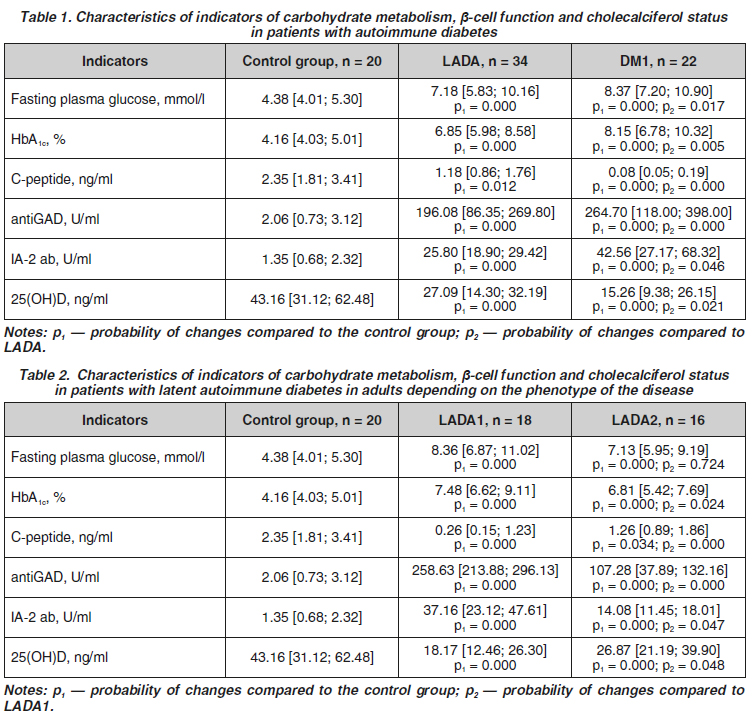
Актуальність. Останнім часом з’явилося чимало відомостей щодо ролі дефіциту холекальциферолу в розвитку класичних типів цукрового діабету (ЦД) та його ускладнень, проте даних відносно латентного автоімунного діабету дорослих (LADA) на сьогодні практично немає. Мета: визначити вплив дефіциту холекальциферолу на компенсацію вуглеводного обміну при LADA. Матеріали та методи. У дослідження включено 56 пацієнтів із ЦД: 34 з LADA та 22 із класичним ЦД 1-го типу (ЦД1), а також 20 практично здорових осіб контрольної групи. Хворих із LADA відповідно до основних фенотипів було поділено на 2 групи: LADA1 та LADA2. Статус холекальциферолу визначали імунохемілюмінесцентним методом. Результати. Показник глікемії натще в групі LADA та ЦД1 був вірогідно вищим, ніж у контролі, — на 63,9 та 91,1 % (p < 0,001) відповідно, також він був вищим на 16,6 % при порівнянні груп ЦД1/LADA (p < 0,05). Рівень HbА1с у 66,1 % пацієнтів дослідних груп становив більше 7 %, що вказує на недостатню компенсацію захворювання. Уміст холекальциферолу був вірогідно нижчим у дослідних групах порівняно з контрольною; при порівнянні LADA/ЦД1 — на 43,7 % нижчим у разі класичного ЦД1 (p < 0,05). Компенсація вуглеводного обміну є гіршою в пацієнтів із LADA1, ніж при LADA2, а найнижчий рівень холекальциферолу реєструвався при LADA1. За даними лінійно-регресійного аналізу кореляцій у пацієнтів із LADA, зворотні зв’язки середньої сили реєструвались між рівнем холекальциферолу та глікемією натще (r = –0,487; p < 0,05), показником HbА1с (r = –0,593; p < 0,05); позитивний — між показником холекальциферолу та С‑пептиду (r = 0,412; p < 0,05). Висновки. У пацієнтів з автоімунним ЦД відзначається недостатнє забезпечення холекальциферолом. Низький уміст холекальциферолу спостерігається в обох групах пацієнтів із LADA незалежно від фенотипу захворювання і асоціюється з гіршими показниками компенсації ЦД.
Background. Recently, a lot of information has appeared on the role of cholecalciferol deficiency in the development of the mechanisms of classical types of diabetes mellitus (DM) and its complications. However, there are currently almost no data regarding latent autoimmune diabetes in adults (LADA). The purpose of this study was to determine the effect of vitamin D deficiency on the compensation of carbohydrate metabolism in LADA. Materials and methods. The study included 56 patients with DM: 34 with LADA and 22 with classical type 1 DM (DM1), as well as 20 practically healthy individuals of the control group. According to the main phenotypes, patients with LADA were divided into 2 groups: LADA1 and LADA2. Cholecalciferol status was determined by the immunochemiluminescence method. Results. The fasting blood glucose level in LADA and DM1 group was significantly higher than in the controls, by 63.9 and 91.1 % (p < 0.001), respectively, and was also 16.6 % higher when comparing DM1/LADA groups (p < 0.05). The level of HbA1c in 66.1 % of patients of the experimental groups was more than 7 %, which indicates insufficient compensation of the disease. The content of vitamin D was significantly lower in the experimental groups compared to the controls; when comparing LADA/DM1 — by 43.7 % lower in case of classical DM1 (p < 0.05). Compensation of carbohydrate metabolism is worse in patients with LADA1 than in LADA2, and the lowest level of vitamin D was recorded in LADA1. According to the linear regression analysis of correlations in patients with LADA, negative correlations of medium strength were recorded between the level of cholecalciferol and fasting blood glucose (r = 0.487; p < 0.05), HbA1c (r = –0.593; p < 0.05); positive — between cholecalciferol and C-peptide (r = 0.412; p < 0.05). Conclusions. In patients with autoimmune diabetes, there is an insufficient supply of cholecalciferol. Low cholecalciferol content is observed in both groups of patients with LADA regardless of the disease phenotype and is associated with worse compensation of DM.
цукровий діабет 1-го типу; латентний автоімунний діабет дорослих; холекальциферол; фенотипи
type 1 diabetes mellitus; latent autoimmune diabetes in adults; cholecalciferol, phenotypes
Introduction
Materials and methods
Results
Discussion
Conclusion
- Jones A.G., McDonald T.J., Shields B.M., Hagopian W., Hattersley A.T. Latent Autoimmune Diabetes of Adults (LADA) Is Likely to Represent a Mixed Population of Autoimmune (Type 1) and Nonautoimmune (Type 2) Diabetes. Diabetes Care. 2021 Jun. 44(6). 1243-51. doi: 10.2337/dc20-2834.
- Groop L., Lundgren V., Lyssenko V. et al. and the Botnia Study Group: Gada positivity in relatives of type 2 diabetes or LADA. Diabetes. 2005. 54(2). 160.
- Pozzilli P., Pieralice S. Latent Autoimmune Diabetes in Adults: Current Status and New Horizons. Endocrinol. Metab. (Seoul). 2018. 33(2). 147-59. doi: 10.3803/EnM.2018.33.2.147.
- Buzzetti R., Zampetti S., Maddaloni E. Adult-onset autoimmune diabetes: current knowledge and implications for management. Nat. Rev. Endocrinol. 2017. 13(11). 674-86. doi: 10.1038/nrendo.2017.99.
- Cervin C., Lyssenko V., Bakhtadze E., Lindholm E., Nilsson P., Tuomi T. et al. Genetic similarities between latent autoimmune diabetes in adults, type 1 diabetes, and type 2 diabetes. Diabetes. 2008 May. 57(5). 1433-7. doi: 10.2337/db07-0299.
- Chang Villacreses M.M., Karnchanasorn R., Panjawatanan P., Ou H.Y., Chiu K.C. Letter to the Editor From Chang Villacreses et al.: “Effects of Vitamin D Supplementation on Insulin Sensitivity and Secretion in Prediabetes”. J. Clin. Endocrinol. Metab. 2022 Jun 16. 107(7). e3086-e3087. doi: 10.1210/clinem/dgac257.
- Gao Y., Chen Z., Ma Z. Vitamin D is Positively Associated with Bone Mineral Density Muscle Mass and Negatively with Insulin Resistance in Senile Diabetes Mellitus. Dis. Markers. 2022 Mar 29. 2022. 9231408. doi: 10.1155/2022/9231408.
- Tucker L.A. Serum, Dietary, and Supplemental Vitamin D Levels and Insulin Resistance in 6294 Randomly Selected, Non-Diabetic U.S. Adults. Nutrients. 2022 Apr 28. 14(9). 1844. doi: 10.3390/nu14091844.
- Urbanovych A., Shykula S. Vitamin D and diabetes mellitus. International Journal of Endocrinology (Ukraine). 2022. 18(1). 78-83. doi: 10.22141/2224-0721.18.1.2022.1148.
- Holick M.F. Vitamin D deficiency. N. Engl. J. Med. 2007 Jul 19. 357(3). 266-81. doi: 10.1056/NEJMra070553.
- Gal-Moscovici A., Sprague S.M. Use of vitamin D in chronic kidney disease patients. Kidney Int. 2010 Jul. 78(2). 146-51. doi: 10.1038/ki.2010.113.
- ElSayed N.A., Aleppo G., Aroda V.R. et al., on behalf of the American Diabetes Association. 2. Classification and Diagnosis of Dia–betes: Standards of Care in Diabetes 2023. Diabetes Care. 2023 Jan 1. 46 (Suppl. 1). S19-S40. doi: 10.2337/dc23-S002.
- Richard I.G. Holt, J. Hans DeVries, Amy Hess-Fischl, Irl B. Hirsch, M. Sue Kirkman et al.; The Management of Type 1 Diabetes in Adults. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2021 Nov 1. 44(11). 2589-625. doi: 10.2337/dci21-0043.
- Fourlanos S., Dotta F., Greenbaum C.J., Palmer J.P., Rolandsson O. et al. Latent autoimmune diabetes in adults (LADA) should be less latent. Diabetologia. 2005 Nov. 48(11). 2206-12. doi: 10.1007/s00125-005-1960-7.
- Buzzetti R., Tuomi T., Mauricio D., Pietropaolo M., Zhou Z. et al. Management of Latent Autoimmune Diabetes in Adults: A Consensus Statement From an International Expert Panel. Diabetes. 2020 Oct. 69(10). 2037-2047. doi: 10.2337/dbi20-0017.
- Lu J., Hou X., Zhang L., Hu C., Zhou J. et al. Associations between clinical characteristics and chronic complications in latent autoimmune diabetes in adults and type 2 diabetes. Diabetes Metab. Res. Rev. 2015 May. 31(4). 411-20. doi: 10.1002/dmrr.2626.
- Pludowski P., Takacs I., Boyanov M., Belaya Z., Diaco–nu C.C. et al. Clinical Practice in the Prevention, Diagnosis and Treatment of Vitamin D Deficiency: A Central and Eastern European Expert Consensus Statement. Nutrients. 2022 Apr 2. 14(7). 1483. doi: 10.3390/nu14071483.
- Pittas A.G., Dawson-Hughes B. Vitamin D and diabetes. J. Steroid Biochem. Mol. Biol. 2010 Jul. 121(1–2). 425-9. doi: 10.1016/j.jsbmb.2010.03.042.
- Oh J., Riek A.E., Darwech I., Funai K., Shao J., Chin K. et al. Deletion of macrophage vitamin D receptor promotes insulin resistance and monocyte cholesterol transport to accelerate atherosclerosis in mice. Cell Rep. 2015 Mar 24. 10(11). 1872-86. doi: 10.1016/j.celrep.2015.02.043.
- Pradhan A. Obesity, metabolic syndrome, and type 2 diabetes: inflammatory basis of glucose metabolic disorders. Nutr. Rev. 2007 Dec. 65 (12 Pt 2). S152-6. doi: 10.1111/j.1753-4887.2007.tb00354.x.
- Dong B., Zhou Y., Wang W., Scott J., Kim K. et al. Vitamin D Receptor Activation in Liver Macrophages Ameliorates Hepatic Inflammation, Steatosis, and Insulin Resistance in Mice. Hepatology. 2020 May. 71(5). 1559-74. doi: 10.1002/hep.30937.
- Leung P.S. The Potential Protective Action of Vitamin D in Hepatic Insulin Resistance and Pancreatic Islet Dysfunction in Type 2 Diabetes Mellitus. Nutrients. 2016 Mar 5. 8(3). 147. doi: 10.3390/nu8030147.
- Vaidya A., Williams J.S. The relationship between vitamin D and the renin-angiotensin system in the pathophysiology of hypertension, kidney disease, and diabetes. Metabolism. 2012 Apr. 61(4). 450-8. doi: 10.1016/j.metabol.2011.09.007.