Международный эндокринологический журнал Том 19, №4, 2023
Вернуться к номеру
Особливості клінічного перебігу перехресту бронхіальної астми та хронічного обструктивного захворювання легень із супутнім цукровим діабетом 2-го типу
Авторы: V.O. Halytska, H.Ya. Stupnytska
Bukovinian State Medical University, Chernivtsi, Ukraine
Рубрики: Эндокринология
Разделы: Клинические исследования
Версия для печати
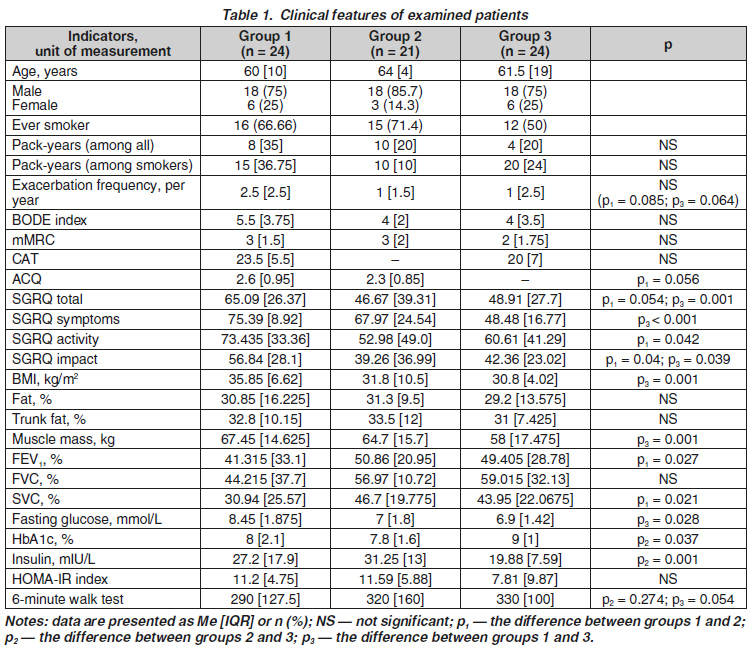
Актуальність. Профілі коморбідності є досить частим предметом вивчення в пацієнтів із перехрестом бронхіальної астми та хронічного обструктивного захворювання легень (ХОЗЛ). Однак у випадку супутнього цукрового діабету 2-го типу (ЦД2) прицільних досліджень щодо якості життя, клінічного перебігу та функції зовнішнього дихання бракує. Мета дослідження: вивчити особливості клінічного перебігу перехресту астми та ХОЗЛ із супутнім ЦД2. Матеріали та методи. Обстежено 69 пацієнтів: 24 — з перехрестом астми та ХОЗЛ і ЦД2 (перша група), 21 особи з астмою та ЦД2 (друга група), 24 — з ХОЗЛ та ЦД2 (третя група). Діагноз перехресту астми та ХОЗЛ встановлювали згідно з рекомендаціями GINA та GOLD (2017). Оцінювали якість життя за опитувальниками САТ, ACQ, SGRQ, вираженість задишки — за шкалою mMRC, тяжкість перебігу та прогнозу захворювання — за індексом BODE. Проведено спірометрію з бронходилатаційним тестом, 6-хвилинний тест ходьби та біоімпедансний аналіз. Результати. Пацієнти основної групи отримали вищу загальну оцінку SGRQ, ніж пацієнти третьої групи (на 33 %, р = 0,001). Вищий показник ACQ та загальна оцінка SGRQ свідчать про тенденцію до гіршого контролю астми та нижчу якість життя в пацієнтів із перехрестом астми й ХОЗЛ та ЦД2 порівняно з групою астми й ЦД2 (р = 0,056 і р = 0,054 відповідно). Індекс маси тіла був вищим, ніж у пацієнтів із ХОЗЛ та ЦД2 (на 16,3 %, р = 0,001). Виявлено вищий уміст глюкози у сироватці крові натще, ніж у пацієнтів із ХОЗЛ та ЦД2 (на 18,3 %, p = 0,028). Об’єм форсованого видиху за першу секунду у групі перехресту астми й ХОЗЛ та ЦД2 був нижчим, ніж при астмі та ЦД2 (на 18,7 %, p = 0,027), а повільна життєва ємність легень зменшилася на 33 % (p = 0,021). Виявлена тенденція до нижчого результату 6-хвилинного тесту ходьби в основній групі порівняно з третьою (р = 0,0548) та вищої частоти загострень за рік, ніж у другій (р = 0,08) та третій групах (р = 0,06). Висновки. У пацієнтів із перехрестом астми та ХОЗЛ і супутнім цукровим діабетом 2-го типу спостерігаються гірші параметри якості життя, нижчий об’єм форсованого видиху за першу секунду та менша повільна життєва ємність легень, субмаксимальна переносимість фізичного навантаження, вищі показники глюкози натще, а також тенденція до збільшення частоти загострень.
Background. Comorbidity profiles are a common subject of research in patients with asthma-COPD (chronic obstructive pulmonary disease) overlap (ACO), but in case of concurrent type 2 diabetes mellitus (T2DM), there is a lack of targeted research on the quality of life, clinical course, and lung function. The aim of the study was to clarify the clinical features of asthma-COPD overlap in combination with T2DM. Materials and methods. Sixty-nine patients were examined: 24 with ACO and T2DM (group 1), 21 with asthma and T2DM (group 2), and 24 with COPD and T2DM (group 3). A diagnosis of ACO was made according to GINA and GOLD 2017 guidelines. Quality of life was assessed using the CAT, ACQ, and SGRQ, and the severity of dyspnea was assessed using the mMRC scale, disease severity and prognosis using the BODE index. Spirometry with bronchodilation test, 6-minute walk test, and bioimpedance analysis were performed. Results. Patients in the main group had a higher total SGRQ score than those in group 3 (by 33 %, p = 0.001). Higher ACQ and total SGRQ scores indicate a trend toward worse asthma control and lower quality of life in patients with ACO and T2DM compared to the asthma + T2DM group (p = 0.056 and p = 0.054, respectively). Body mass index was higher than in patients with COPD and T2DM (by 16.3 %, p = 0.001). Higher serum glucose levels were found in patients with ACO and T2DM than in those with COPD and T2DM (by 18.3 %, p = 0.028). The FEV1 in the ACO and T2DM group was lower than in the asthma + T2DM group (by 18.7 %, p = 0.027), and the SVC was lower by 33 % (p = 0.021). There was a tendency to a lower result in the 6-minute walk test in the main group compared to patients from group 3 (p = 0.0548), and a higher frequency of exacerbations per year compared to groups 2 (p = 0.08) and 3 (p = 0.06). Conclusions. Patients with asthma-COPD overlap and concurrent type 2 diabetes mellitus have worse quality of life, lower FEV1 and SVC, submaximal exercise tolerance, higher fasting glucose levels, and a tendency towards increased exacerbation frequency.
бронхіальна астма; хронічне обструктивне захворювання легень; перехрест астми та ХОЗЛ; цукровий діабет 2-го типу; коморбідність; якість життя; функція зовнішнього дихання
asthma; chronic obstructive pulmonary disease; asthma-COPD overlap; type 2 diabetes mellitus; comorbidity; quality of life; pulmonary function
Introduction
Materials and methods
Results
Discussion
Conclusions
- Global Initiative for Asthma. Global Strategy for Asthma Mana–gement and Prevention, 2022. Available from: https://ginasthma.org.
- Global Initiative for Asthma (GINA). Diagnosis and initial treatment of asthma, COPD, and asthma-COPD overlap: a joint pro–ject of GINA and GOLD updated April 2017. Available from: https://ginasthma.org/wp-content/uploads/2019/11/GINA-GOLD-2017-overlap-pocket-guide-wms-2017-ACO.pdf.
- Toledo-Pons N., van Boven J.F.M., Román-Rodríguez M. et al. ACO: time to move from the description of different phenotypes to the treatable traits. PLoS ONE. 2019 Jan 24. 14(1). e0210915. doi: 10.1371/journal.pone.0210915.
- Lim J.U., Kim D.K., Lee M.G. et al. Clinical Characteristics and Changes of Clinical Features in Patients with Asthma-COPD Overlap in Korea according to Different Diagnostic Criteria. Tuberc. Respir. Dis. 2020. 83(1). 34-45. doi: 10.4046/trd.2020.0031.
- Barrecheguren M., Pinto L., Mostafavi-Pour-Manshadi S.-M. et al. Identification and definition of asthma-COPD overlap: the CanCOLD study. Respirology. 2020. 25. 836-49. doi: 10.1111/resp.13780.
- Roman-Rodriguez M., Kaplan A. GOLD 2021 Strategy Report: Implications for Asthma-COPD Overlap. Int. J. Chron. Obstruct. Pulmon. Dis. 2021 Jun 14. 16. 1709-15. doi: 10.2147/COPD.S300902.
- Romem A., Rokach A., Bohadana A. et al. Identification of Asthma-COPD Overlap, Asthma, and Chronic Obstructive Pulmonary Disease Phenotypes in Patients with Airway Obstruction: Influence on Treatment Approach. Respiration. 2020. 99(1). 35-42. doi: 10.1159/000503328.
- Hosseini M., Almasi-Hashiani A., Sepidarkish M., Maroufizadeh S. Global prevalence of asthma-COPD overlap (ACO) in the general population: a systematic review and meta-analysis. Respir. Res. 2019 Oct 23. 20(1). 229. doi: 10.1186/s12931-019-1198-4.
- Dash R.R., Panda B., Panigrahi M., Nayak B. A Step Toward the Exploration of Better Spirometric Parameters for Early Diagnosis of Pulmonary Dysfunction in Persons With Type 2 Diabetes Mellitus. Cureus. 2022 Jul 6. 14(7). e26622. doi: 10.7759/cureus.26622.
- Maselli D.J., Hanania N.A. Asthma COPD overlap: impact of associated comorbidities. Pulm. Pharmacol. Ther. 2018 Oct. 52. 27-31. doi: 10.1016/j.pupt.2018.08.006.
- Peltola L., Pätsi H., Harju T. COPD Comorbidities Predict High Mortality — Asthma-COPD-Overlap Has Better Prognosis. COPD: Journal of Chronic Obstructive Pulmonary Disease. 2020. 17(4). 366-72. doi: 10.1080/15412555.2020.1783647.
- Akmatov M.K., Ermakova T., Holstiege J., Steffen A., von Stillfried D., Bätzing J. Comorbidity profile of patients with concurrent diagnoses of asthma and COPD in Germany. Sci. Rep. 2020 Oct 21. 10(1). 17945. doi: 10.1038/s41598-020-74966-1.
- Tianshi D.W. Diabetes and Glycemic Dysfunction in Asthma. J. Allergy Clin. Immunol. Pract. 2020. 8. 3416-7. doi: 10.1016/j.jaip.2020.07.011.
- Yang G., Han Y.Y., Forno E. et al. Glycated Hemoglobin A1c, Lung Function, and Hospitalizations Among Adults with Asthma. J. Allergy Clin. Immunol. Pract. 2020 Nov-Dec. 8(10). 3409-15.e1 doi: 10.1016/j.jaip.2020.06.017.
- Wu T.D., Brigham E.P., Keet C.A., Brown T.T., Hansel N.N., McCormack M.C. Association Between Prediabetes/Diabetes and Asthma Exacerbations in a Claims-Based Obese Asthma Cohort. J. Allergy Clin. Immunol. Pract. 2019. 7(6). 1868-73.e5 doi: 10.1016/j.jaip.2019.02.029.
- Santos N.C.D., Miravitlles M., Camelier A.A., Almeida V.D.C., Maciel R.R.B.T., Camelier F.W.R. Prevalence and Impact of Comorbidities in Individuals with Chronic Obstructive Pulmonary Disease: A Systematic Review. Tuberc. Respir. Dis. (Seoul). 2022 Jul. 85(3). 205-20. doi: 10.4046/trd.2021.0179.
- Shah C.H., Reed R.M., Liang Y., Zafari Z. Association bet–ween lung function and future risks of diabetes, asthma, myocardial infarction, hypertension and all-cause mortality. ERJ Open Res. 2021. 7. 00178-2021. doi: 10.1183/23120541.00178-2021.
- Peerboom S., Graff S., Seidel L. et al. Predictors of a good response to inhaled corticosteroids in obesity-associated asthma. Biochem. Pharmacol. 2020 Sep. 179. 113994. doi: 10.1016/j.bcp.2020.113994.
- Zysman M., Deslee G., Perez T. et al. Burden and Characte–ristics of Severe Chronic Hypoxemia in a Real-World Cohort of Subjects with COPD. Int. J. Chron. Obstruct. Pulmon. Dis. 2021 May 10. 16. 1275-84. doi: 10.2147/COPD.S295381.
- Peng J., Wang M., Wu Y., Shen Y., Chen L. Clinical Indicators for Asthma-COPD Overlap: A Systematic Review and Meta-Ana–lysis. Int. J. Chron. Obstruct. Pulmon. Dis. 2022 Oct 12. 17. 2567-75. doi: 10.2147/COPD.S374079.
- Izbicki G., Teo V., Liang J. et al. Clinical Characteristics of Patients With Asthma COPD Overlap (ACO) in Australian Primary Care. Int. J. Chron. Obstruct. Pulmon. Dis. 2019. 14. 2745-52. doi: 10.2147/COPD.S220346.
- An T.J., Rhee C.K., Park Y.B., Yoo K.H., Yoon H.K. FVC, but not FEV1, is associated with clinical outcomes of asthma-COPD overlap. Sci. Rep. 2022 Aug 15. 12(1). 13820. doi: 10.1038/s41598-022-15612-w.
- Sánchez Castillo S., Smith L., Díaz Suárez A., López Sánchez G.F. Limitations in Activities of Daily Living among Older Adults with COPD, Asthma, or Asthma-COPD Overlap Residing in Spain. Int. J. Environ. Res. Public Health. 2023. 20(4). 3467. doi: 10.3390/ijerph20043467.
- Uppal P., Mohammed S.A., Rajashekar S. et al. Type 2 Dia–betes Mellitus and Asthma: Pathomechanisms of Their Association and Clinical Implications. Cureus. 2023. 15(3). e36047. doi: 10.7759/cureus.36047.
- Kopf S., Kumar V., Kender Z. et al. Diabetic Pneumopathy — A New Diabetes-Associated Complication: Mechanisms, Consequences and Treatment Considerations. Front. Endocrinol. 2021. 12. 765201. doi: 10.3389/fendo.2021.765201.
- Grasemann H., Holguin F. Oxidative stress and obesity-rela–ted asthma. Paediatr. Respir. Rev. 2021 Mar. 37. 18-21. doi: 10.1016/j.prrv.2020.05.004.
- Ghosh N., Choudhury P., Kaushik S.R. et al. Metabolomic fingerprinting and systemic inflammatory profiling of asthma COPD overlap (ACO). Respir. Res. 2020. 21. 126. doi: 10.1186/s12931-020-01390-4.
- Nambiar S., Bong How S., Gummer J., Trengove R., Moodley Y. Metabolomics in chronic lung diseases. Respirology. 2020. 25. 139-48. doi: 10.1111/resp.13530.
- Ghosh N., Choudhury P., Joshi M. et al. Global metabolome profiling of exhaled breath condensates in male smokers with asthma COPD overlap and prediction of the disease. Sci. Rep. 2021. 11. 16664. doi: 10.1038/s41598-021-96128-7.
- Duong K.N.C., Tan C.J., Rattanasiri S., Thakkinstian A., Anothaisintawee T., Chaiyakunapruk N. Comparison of diagnostic accuracy for diabetes diagnosis: a systematic review and network meta-analysis. Front. Med. (Lausanne). 2023 Jan 24. 10. 1016381. doi: 10.3389/fmed.2023.1016381.
- Puri D., Kaur J., Gaur N., Kodidala S.R. Role of glycated hemoglobin in microvascular complications in type 2 dia–betes mellitus: cross sectional study. International Journal of Endocrinology (Ukraine). 2022. 18(6). 319-323. doi: 10.22141/2224-0721.18.6.2022.1201.
- Ceriello A., Prattichizzo F., Phillip M., Hirsch I.B., Ma–thieu C., Battelino T. Glycaemic management in diabetes: old and new approaches. Lancet Diabetes Endocrinol. 2022 Jan. 10(1). 75-84. doi: 10.1016/S2213-8587(21)00245-X.
- Ceriello A. Glucose Variability and Diabetic Complications: Is It Time to Treat? Diabetes Care. 2020 Jun. 43(6). 1169-1171. doi: 10.2337/dci20-0012.