Международный эндокринологический журнал Том 19, №4, 2023
Вернуться к номеру
Папілярна карцинома щитоподібної залози на тлі доброякісної тиреоїдної та паратиреоїдної патології: клінічні й патоморфологічні особливості
Авторы: A. Dinets (1–3), M. Gorobeiko (1), V. Hoperia (1), A. Lovin (1), S. Tarasenko (4)
(1) — Institute of Biology and Medicine, Taras Shevchenko National University of Kyiv, Kyiv, Ukraine
(2) — National Scientific Center “Institute of Beekeeping named after P.I. Prokopovich”, Kyiv, Ukraine
(3) — Kyiv Agrarian University, Kyiv, Ukraine
(4) — EuroMedCare Hospital St. Anna’s, Piaseczno, Poland
Рубрики: Эндокринология
Разделы: Клинические исследования
Версия для печати
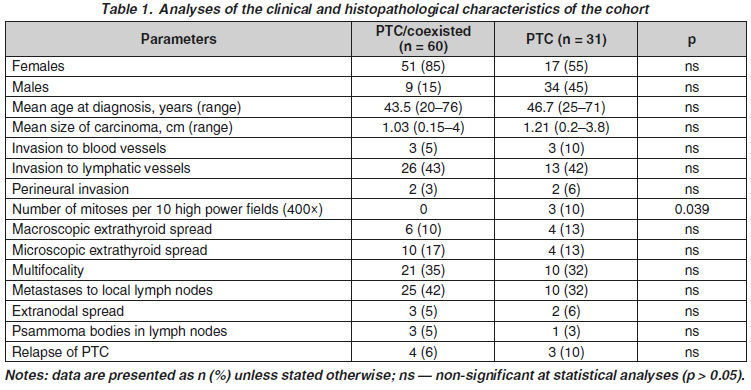
Актуальність. Папілярна карцинома щитоподібної залози (ПКЩЗ) є найбільш частим злоякісним новоутворенням, що походить із фолікулярних клітин щитоподібної залози. ПКЩЗ часто діагностується на тлі іншої доброякісної патології щитоподібної залози: хронічного лімфоцитарного тиреоїдиту, фолікулярної аденоми щитоподібної залози, токсичної аденоми щитоподібної залози, хвороби Грейвса (дифузний токсичний зоб), нетоксичного багатовузлового зоба. Мета: оцінити поширеність та визначити відмінності клініко-патоморфологічних показників ПКЩЗ на тлі іншої доброякісної патології щитоподібної залози. Матеріали та методи. Ідентифіковано 91 хворого з ПКЩЗ, які проходили оперативне лікування на клінічних базах кафедри хірургії ННЦ «Інститут біології та медицини» Національного університету імені Тараса Шевченка (Київ). Результати. У досліджуваній групі ПКЩЗ 31 (35 %) пацієнт не мав супутньої доброякісної патології щитоподібної залози та 60 (35 %) пацієнтів мали. Статистично вірогідно вищу частоту мітозів на 10 полів зору великого збільшення (400×) виявлено в 3 (10 %) пацієнтів із ПКЩЗ порівняно з відсутністю цього патогістологічного параметра в осіб із ПКЩЗ на тлі іншої патології (р = 0,037). Подальший аналіз показав, що 4 мітотичні фігури були знайдені в одному з цих трьох випадків ПКЩЗ і один мітоз — в інших двох випадках на 10 полів зору при великому збільшенні (400×). Висновки. Папілярна карцинома щитоподібної залози на тлі іншої доброякісної патології щитоподібної залози асоціюється з нижчою біологічною агресивністю, ніж ПКЩЗ без супутньої патології, про що свідчить менша кількість мітотичних фігур на 10 полів зору великого збільшення. Наявність супутньої доброякісної патології щитоподібної залози можна розглядати як сприятливий прогностичний фактор для хворих на ПКЩЗ.
Background. Papillary thyroid carcinoma (PTC) is the most frequent malignant neoplasm originating from follicular cells of thyroid gland. PTC is often coexisting with other benign thyroid pathology such as chronic lymphocytic thyroiditis, follicular thyroid adenoma, toxic thyroid adenoma, Graves’ disease, non-toxic multinodular goiter. PTC can be diagnosed in patients undergoing surgical treatment for hyperparathyroidism. However, the clinical and pathomorphological features of PTC in the presence of other benign thyroid or parathyroid pathology, as well as possible differences in surgical tactics, remain relevant as evidenced by previously published reports. The purpose of the study is to assess the prevalence and determine the differences in the clinical and pathomorphological parameters of PTC in the presence of benign thyroid and parathyroid pathology. Materials and methods. There were identified 91 patients with PTC, who underwent surgical treatment at the clinical bases of the Department of Surgery of the NSC “Institute of Biology and Medicine” of Taras Shevchenko National University of Kyiv. Results. In the studied PTC cohort, 31 (35 %) patients were without coexisted benign thyroid pathology and 60 (35 %) patients had it (PTC/coexisted). A statistically significantly higher number of mitoses per 10 high power fields in the PTC group were identified in 3 (10 %) patients, as compared to the absence of this pathohistological parameter in the PTC/coexisted (p = 0.037). Further analysis of these 3 cases showed that 4 mitotic figures were found in one PTC, and one mitotic figure in the other two cases per 10 high power fields (400×). Conclusions. Papillary thyroid carcinoma in patients with other benign thyroid pathology is associated with lower biological aggressiveness than PTC without comorbidity as evidenced by a lower frequency of mitotic figures per 10 high power fields. The presence of concomitant benign thyroid pathology can be considered as a favorable prognostic factor for patients with PTC.
папілярна карцинома щитоподібної залози; супутня тиреоїдна патологія; фігури мітозів на 10 полів зору великого збільшення (400×)
papillary thyroid carcinoma; accompanying thyroid pathology; mitoses per 10 high power fields (400×)
Introduction
Materials and methods
Results
Discussion
Conclusions
- Ito Y., Miyauchi A., Kihara M., Fukushima M., Higashiyama T., Miya A. Overall Survival of Papillary Thyroid Carcinoma Patients: A Sing–le-Institution Long-Term Follow-Up of 5897 Patients. World J. Surg. 2018 Mar. 42(3). 615-622. doi: 10.1007/s00268-018-4479-z.
- Nabhan F., Dedhia P.H., Ringel M.D. Thyroid cancer, recent advances in diagnosis and therapy. Int. J. Cancer. 2021 Sep 1. 149(5). 984-992. doi: 10.1002/ijc.33690.
- Prete A., Borges de Souza P., Censi S., Muzza M., Nucci N., Sponziello M. Update on Fundamental Mechanisms of Thyroid Cancer. Front. Endocrinol. (Lausanne). 2020 Mar 13. 11. 102. doi: 10.3389/fendo.2020.00102.
- Xu S., Huang H., Qian J., Liu Y., Huang Y. et al. Prevalence of Hashimoto Thyroiditis in Adults With Papillary Thyroid Cancer and Its Association With Cancer Recurrence and Outcomes. JAMA Netw. Open. 2021 Jul 1. 4(7). e2118526. doi: 10.1001/jamanetworkopen.2021.18526.
- Varadharajan K., Choudhury N. A systematic review of the incidence of thyroid carcinoma in patients undergoing thyroidectomy for thyrotoxicosis. Clin. Otolaryngol. 2020 Jul. 45(4). 538-544. doi: 10.1111/coa.13527.
- Rosario P.W., de Castro Nicolau T. Report of one case of malignancy among 17 autonomous thyroid nodules in children and adolescents. J. Paediatr. Child Health. 2021 Jun. 57(6). 810-812. doi: 10.1111/jpc.15322.
- Brito J.P., Hay I.D. Management of Papillary Thyroid Microcarcinoma. Endocrinol. Metab. Clin. North Am. 2019 Mar. 48(1). 199-213. doi: 10.1016/j.ecl.2018.10.006.
- Cibas E.S., Ali S.Z. The 2017 Bethesda System for Reporting Thyroid Cytopathology. Thyroid. 2017 Nov. 27(11). 1341-1346. doi: 10.1089/thy.2017.0500.
- Hajeer M.H., Awad H.A., Abdullah N.I., Almuhaisen G.H., Abudalu L.E. The rising trend in papillary thyroid carcinoma. True increase or over diagnosis? Saudi Med. J. 2018 Feb. 39(2). 147-153. doi: 10.15537/smj.2018.2.21211.
- Baloch Z.W., Asa S.L., Barletta J.A., Ghossein R.A., Juhlin C.C. et al. Overview of the 2022 WHO Classification of Thyroid Neoplasms. Endocr. Pathol. 2022 Mar. 33(1). 27-63. doi: 10.1007/s12022-022-09707-3.
- Jeong C., Kwon H.I., Baek H., Kim H.S., Lim D.J. et al. Association of Hyperparathyroidism and Papillary Thyroid Cancer: A Multicenter Retrospective Study. Endocrinol. Metab. (Seoul). 2020 Dec. 35(4). 925-932. doi: 10.3803/EnM.2020.725.
- Haugen B.R., Alexander E.K., Bible K.C., Doherty G.M., Mandel S.J. et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016 Jan. 26(1). 1-133. doi: 10.1089/thy.2015.0020.
- Basolo F., Macerola E., Poma A.M., Torregrossa L. The 5th edition of WHO classification of tumors of endocrine organs: changes in the diagnosis of follicular-derived thyroid carcinoma. Endocrine. 2023 Jun. 80(3). 470-476. doi: 10.1007/s12020-023-03336-4.
- Gorobeiko M., Dinets A., Pominchuk D., Abdalla K., Prylutskyy Y., Hoperia V. Challenges of Differential Diagnosis Between Primary Hyperparathyroidism and Bone Metastases of Breast Cancer. Clin. Med. Insights Case Rep. 2022. 15. 11795476221125136.
- Gorobeiko M., Dinets A., Hoperia V., Abdalla K. Improved intraoperative verification of parathyroid glands by determining their autofluorescence in the infrared spectrum. International Journal of Endocrinology (Ukraine). 2021. 17(6). 465-471. doi: 10.22141/2224-0721.17.6.2021.243207.
- Tong J., Ruan M., Jin Y., Fu H., Cheng L. et al. Poorly diffe–rentiated thyroid carcinoma: a clinician’s perspective. Eur. Thyroid J. 2022 Mar 24. 11(2). e220021. doi: 10.1530/ETJ-22-0021.
- Xu B., Ghossein R. Poorly differentiated thyroid carcinoma. Semin. Diagn. Pathol. 2020 Sep. 37(5). 243-247. doi: 10.1053/j.semdp.2020.03.003.
- Wong K.S., Dong F., Telatar M., Lorch J.H., Alexander E.K. et al. Papillary Thyroid Carcinoma with High-Grade Features Versus Poorly Differentiated Thyroid Carcinoma: An Analysis of Clinicopathologic and Molecular Features and Outcome. Thyroid. 2021 Jun. 31(6). 933-940. doi: 10.1089/thy.2020.0668.
- Palamarchuk A., Vlasenko M., Prudius P., Kolomiets V. Epidemiology and Morphological Features of Thyroid Cancer in People Living in Vinnytsia Region. International Journal of Endocrinology (Ukraine). 2016. 7(79). 82-85. doi: 10.22141/2224-0721.7.79.2016.86423.