Международный эндокринологический журнал Том 19, №4, 2023
Вернуться к номеру
Аспекти розвитку гіпотиреоїдної кардіоміопатії на фоні хронічного системного запалення
Авторы: О.Yu. Horodynska, О.V. Muravlova, Z.О. Shaienko, І.L. Dvornyk
Poltava State Medical University, Poltava, Ukraine
Рубрики: Эндокринология
Разделы: Клинические исследования
Версия для печати
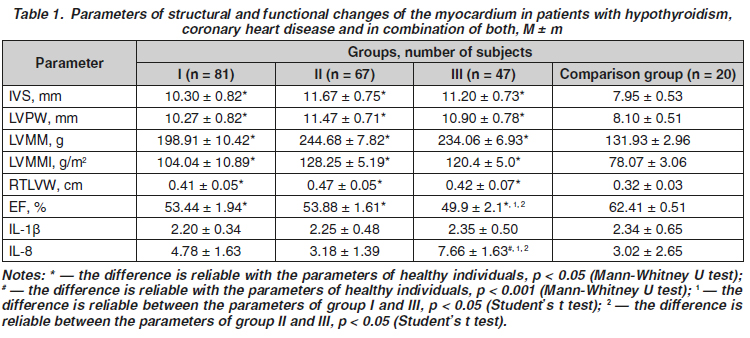
Актуальність. Серцево-судинні прояви посідають одне з перших місць у симптоматиці гіпотиреозу. Оскільки поширеність як ішемічної хвороби серця (ІХС), так і гіпотиреозу найбільша у віковій групі понад 50 років, зростає проблема поєднання цих нозологій в осіб старшого віку. Мета дослідження: вивчити структурно-функціональний стан міокарда лівого шлуночка при гіпотиреозі та ІХС на фоні хронічного системного запалення. Матеріали та методи. Відповідно до поставлених завдань було проведене рандомізоване контрольоване клінічне дослідження в паралельних групах пацієнтів із гіпотиреозом, ІХС та за умов поєднаної патології. Для формування основної групи виконано скринінговий аналіз 556 історій хвороби осіб із гіпотиреозом та ІХС протягом 2006–2015 років, що були відібрані для подальшого вивчення. Результати. Виявлено, що гіпертрофія міокарда розвивається в усіх групах хворих, зниження функції щитоподібної залози характеризується ремоделюванням міокарда лівого шлуночка з розвитком ексцентричної гіпертрофії, прогресуванням систолічної дисфункції в умовах коморбідності. Знайдений прямий кореляційний зв’язок зниження фракції викиду з підвищенням рівня інтерлейкіну-8. При оцінці хронічного системного запалення виявлено достовірне підвищення рівня інтерлейкіну-8 у пацієнтів з ІХС, асоційованою з гіпотиреозом (7,66 ± 2,18 пг/мл; р < 0,05). Це свідчить про те, що збереження прозапального стану в пацієнтів із поєднаною патологією є негативним прогностичним фактором розвитку серцево-судинних ускладнень. Висновки. При ехокардіоскопії хворих на гіпотиреоз порушення центральної гемодинаміки можна визначити за показниками діастолічної та систолічної функції серця. В осіб з ізольованим гіпотиреозом та при його поєднанні з ішемічною хворобою серця відзначається потовщення стінок міокарда лівого шлуночка, що свідчить про специфічність змін геометрії серця і призводить до розвитку ексцентричної гіпертрофії. Ці показники можна вважати маркером ураження серця при гіпотиреозі, вираженість якого може визначити тяжкість гіпотиреозу. Активація хронічного системного запалення більш виражена за умов коморбідності, з негативним прогностичним впливом на стан серцево-судинної системи.
Background. Cardiovascular manifestations are rated first among the symptoms of hypothyroidism. Since the highest prevalence of both coronary heart disease (CHD) and hypothyroidism is observed in the age group over 50 years old, the problem of combination of these nosologies in older people is increasing. Aim of the study is to investigate the structural and functional state of the left ventricular myocardium in hypothyroidism and CHD associated with chronic systemic inflammation. Materials and methods. To reach the objectives of the study, a randomized controlled clinical trial has been conducted in parallel groups of patients with hypothyroidism, CHD and combination of both. To form the main group, a screening analysis of 556 medical histories of patients with hypothyroidism and CHD during the period of 2006–2015 has been made, which were selected for further study. Results. It has been found that myocardial hypertrophy develops in all groups of patients, a decrease in thyroid function leads to remodeling of the left ventricular myocardium with the development of eccentric hypertrophy and the progression of systolic heart failure in comorbidity. A direct correlation between reduced ejection fraction and elevated interleukin-8 level has been detected. Evaluation of the state of chronic systemic inflammation revealed a significant increase in the level of interleukin-8 in patients with coronary heart disease associated with hypothyroidism (7.66 ± 2.18 pg/ml; p < 0.05). This indicate that the persistence of pro-inflammatory state in patients with combined pathology is a negative prognostic factor for the development of cardiovascular complications. Conclusion. During echocardioscopy of patients with hypothyroidism, the impaired central hemodynamics can be determined by the indicators of the diastolic and systolic heart function. In patients with isolated hypothyroidism and in combination with coronary heart disease, thickening of the left ventricular myocardial walls is noted, which proves the specificity of changes in the heart geometry that leads to the development of eccentric hypertrophy. This can be considered as the marker of a “hypothyroid” heart whose severity can determine the severity of hypothyroidism. Activation of chronic systemic inflammation is more pronounced in conditions of comorbidity, with a negative prognostic effect on the state of the cardiovascular system.
щитоподібна залоза; серце; гіпотиреоз; гіпотиреоїдна кардіоміопатія; гіпертрофія міокарда; прозапальні цитокіни
thyroid gland; heart; hypothyroidism; hypothyroid cardiomyopathy; myocardial hypertrophy; proinflammatory cytokines
Introduction
Materials and methods
Results
Discussion
Conclusions
- Wilson S.A., Stem L.A., Bruehlman R.D. Hypothyroidism: Diagnosis and Treatment. Am. Fam. Physician. 2021 May 15. 103(10). 605-613.
- Horodynska O. Syndrome of hypothyroidism as a factor of cardiovascular pathology development (literature review). Internatio–nal Journal of Endocrinology (Ukraine). 2017. 13(7). 503-505. doi: 10.22141/2224-0721.13.7.2017.115749.
- Malin A.J., Riddell J., McCague H., Till C. Fluoride exposure and thyroid function among adults living in Canada: effect modification by iodine status. Environ. Int. 2018 Dec. 121(Pt 1). 667-674. doi: 10.1016/j.envint.2018.09.026.
- Wang M., Liu L., Li H., Li Y., Liu H. et al. Thyroid function, intelligence, and low-moderate fluoride exposure among Chinese school-age children. Environ. Int. 2020 Jan. 134. 105229. doi: 10.1016/j.envint.2019.105229.
- Malakar A.K., Choudhury D., Halder B., Paul P., Uddin A., Chakraborty S. A review on coronary artery disease, its risk factors, and therapeutics. J. Cell. Physiol. 2019 Aug. 234(10). 16812-16823. doi: 10.1002/jcp.28350.
- Parel P.M., Berg A.R., Hong C.G., Florida E.M., O’Hagan R. et al. Updates in the Impact of Chronic Systemic Inflammation on Vascular Inflammation by Positron Emission Tomography (PET). Curr. Cardiol. Rep. 2022 Apr. 24(4). 317-326. doi: 10.1007/s11886-022-01651-2.
- Furman D., Campisi J., Verdin E., Carrera-Bastos P., Targ S. et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019 Dec. 25(12). 1822-1832. doi: 10.1038/s41591-019-0675-0.
- Mundula T., Russo E., Curini L., Giudici F., Piccioni A. et al. Chronic Systemic Low-Grade Inflammation and Modern Lifestyle: The Dark Role of Gut Microbiota on Related Diseases with a Focus on COVID-19 Pandemic. Curr. Med. Chem. 2022. 29(33). 5370-5396. doi: 10.2174/0929867329666220430131018.
- Chang C.Y., Chien Y.J., Lin P.C., Chen C.S., Wu M.Y. Nonthyroidal Illness Syndrome and Hypothyroidism in Ischemic Heart Disease Population: A Systematic Review and Meta-Analysis. J. Clin. Endocrinol. Metab. 2020 Aug 1. 105(8). dgaa310. doi: 10.1210/clinem/dgaa310.
- Martinez S.S., Gutierrez M., Delgado-Enciso I., Maisonet J., Pierre A.J. et al. Economic and Cardiometabolic Risk Factors Are Predictors of Lower Thyroid Stimulating Hormone (TSH) Levels in Hispanic/Latinx Adults with Euthyroidism — A Community-Based Study. Int. J. Environ. Res. Public Health. 2022 Jul 2. 19(13). 8142. doi: 10.3390/ijerph19138142.
- Udovcic M., Pena R.H., Patham B., Tabatabai L., Kansara A. Hypothyroidism and the Heart. Methodist DeBakey Cardiovasc. J. 2017 Apr-Jun. 13(2). 55-59. doi: 10.14797/mdcj-13-2-55.
- Abdel-Moneim A., Gaber A.M., Gouda S., Osama A., Othman S.I., Allam G. Relationship of thyroid dysfunction with cardiovascular diseases: updated review on heart failure progression. Hormones (Athens). 2020 Sep. 19(3). 301-309. doi: 10.1007/s42000-020-00208-8.
- Bielecka-Dabrowa A., Godoy B., Suzuki T., Banach M., von Haehling S. Subclinical hypothyroidism and the development of heart failure: an overview of risk and effects on cardiac function. Clin. Res. Cardiol. 2019 Mar. 108(3). 225-233. doi: 10.1007/s00392-018-1340-1.
- Erge E., Kiziltunc C., Balci S.B., Atak Tel B.M., Bilgin S. et al. A Novel Inflammatory Marker for the Diagnosis of Hashimoto’s Thyroiditis: Platelet-Count-to-Lymphocyte-Count Ratio. Diseases. 2023 Jan 22. 11(1). 15. doi: 10.3390/diseases11010015.
- Gu Y., Wang Y., Zhang Q., Liu L., Meng G. et al. The association between thyroid function and incidence of metabolic syndrome in euthyroid subjects: Tianjin chronic low-grade systemic inflammation and health cohort study. Clin. Endocrinol. (Oxford). 2018 May. 88(5). 735-743. doi: 10.1111/cen.13576.
- Patel A., Zhan Y. Hypothyroidism leading to a syncopal episode: a reminder to check drug interactions/a case of iatrogenic hypothyroidism presented with cardio-inhibitory syncope and resolved by thyroxine supplementation. Anadolu Kardiyol. Derg. 2012 Aug. 12(5). 453-4; author reply 454. doi: 10.5152/akd.2012.140.
- Purdenko T.I., Lytvynenko N.V., Ostrovskaya L.I., Hladka V.M., Sylenko H.Y., Tarianyk K.A. On the problem of syncopal conditions in the neurological practice. Wiad. Lek. 2018. 71(9). 1823-1828 (in Ukrainian).
- Begum L.N., Hoque M.H., Mahmood M., Rahman M., Ahmed S.P., Islam M.S. Echocardiographic Evaluation of Cardiac Changes in Patients with Hypothyroidism and the Response to Treatment. Mymensingh Med. J. 2022 Jul. 31(3). 790-796.
- De Luca R., Davis P.J., Lin H.Y., Gionfra F., Percario Z.A. et al. Thyroid Hormones Interaction With Immune Response, Inflammation and Non-thyroidal Illness Syndrome. Front. Cell Dev. Biol. 2021 Jan 21. 8. 614030. doi: 10.3389/fcell.2020.614030.
- Hadzović-Dzuvo A., Kucukalić-Selimović E., Nakas-Ićin–dić E., Rasić S., Begić A. et al. Echocardiographic evaluation of cardiac function in female patients with thyroid disorders. Bosn. J. Basic Med. Sci. 2010 May. 10(2). 112-5. doi: 10.17305/bjbms.2010.2704.
- Malhotra Y., Kaushik R.M., Kaushik R. Echocardiographic evaluation of left ventricular diastolic dysfunction in subclinical hypothyroidism: a case-control study. Endocr. Res. 2017 Aug. 42(3). 198-208. doi: 10.1080/07435800.2017.1292524.