Международный эндокринологический журнал Том 22, №1, 2026
Вернуться к номеру
Рівень ZEB1 у тканині папілярної карциноми щитоподібної залози, метастазах, мононуклеарних клітинах крові та плазмі
Авторы: P.P. Zinych, V.M. Pushkarev, N.I. Levchuk, Ye.A. Shelkovoy, M.Yu. Bolgov
State Institution “V.P. Komisarenko Institute of Endocrinology and Metabolism of the NAMS of Ukraine”, Kyiv, Ukraine
Рубрики: Эндокринология
Разделы: Клинические исследования
Версия для печати
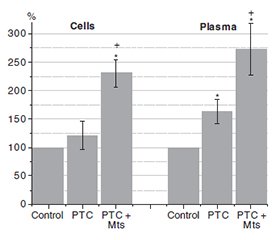
Актуальність. Епітеліально-мезенхімальний перехід (ЕМП) є ключовим процесом у поширенні пухлинних клітин. Білок ZEB1 є центральним транскрипційним регулятором ЕМП — клітинної програми, яка лежить в основі інвазії пухлини, метастатичної дисемінації та терапевтичної резистентності. Дані свідчать про те, що аберантна активація ZEB1 обумовлює епітеліальну пластичність, інвазивну поведінку й метастатичну компетентність клітин раку щитоподібної залози. Мета: порівняти експресію ZEB1 в умовно нормальній тканині щитоподібної залози, доброякісних новоутвореннях, пухлинній тканині папілярної карциноми щитоподібної залози (ПКЩЗ) і метастазах, а також у клітинах і плазмі крові пацієнтів із ПКЩЗ. Матеріали та методи. Плазму крові, мононуклеарні клітини, післяопераційні зразки пухлинної тканини, метастазів та умовно нормальної тканини отримали у хірургічному відділенні клініки інституту. Концентрацію ZEB1 визначали за допомогою наборів для імуноферментного аналізу. Статистичний аналіз і візуалізацію даних здійснювали за допомогою програмного забезпечення Origin 2019b. Результати. Кількість ZEB1 у пухлинній тканині перевищує таку в умовно нормальній (гістологічно незміненій) тканині більше ніж у 5 разів. Приблизно таке співвідношення рівня ZEB1 між нормальною і пухлинною тканинами спостерігається у зразках пацієнтів із ПКЩЗ та метастазами. Концентрація транскрипційних факторів у метастазах перевищує норму майже в 10 разів. Кількість ZEB1 у тканині зоба не відрізняється від такої в умовно нормальній тканині пацієнтів із ПКЩЗ, але суттєво відрізняється від показника пухлинної тканини карциноми. Висновки. Наші дані свідчать про значні відмінності в концентрації ZEB1 між умовно нормальною та пухлинною тканинами щитоподібної залози. Рівень ZEB1 у метастазах був ще вищим. Слід підкреслити той факт, що як у клітинах крові, так і в плазмі пацієнтів із метастатичною ПКЩЗ рівень ZEB1 був значно вищим, ніж у здорових людей та осіб із ПКЩЗ без метастазів. Останнє може стати основою для передопераційного прогнозу розвитку метастазів у пацієнтів із ПКЩЗ.
Background. Epithelial-mesenchymal transition (EMT) is a key process in the dissemination of tumor cells. Zinc finger E-box-binding homeobox 1 (ZEB1) is a central transcriptional regulator of EMT, a cellular program that underlies tumor invasion, metastatic dissemination, and therapeutic resistance. Evidence indicates that aberrant activation of ZEB1 promotes epithelial plasticity, invasive behavior, and metastatic competence of thyroid cancer cells. The purpose of the study was to compare the expression of ZEB1 in conditionally normal thyroid tissue, benign neoplasms, tumor tissue of papillary thyroid carcinoma (PTC) and metastasis, as well as in cells and blood plasma of patients with PTC. Materials and methods. Blood plasma, mononuclear cells, postoperative samples of tumor tissue, metastasis, and conditionally normal tissue were obtained from the surgical department of the Institute’s clinic. The ZEB1 concentration was determined using enzyme immunoassay kits. Statistical analysis and data presentation were performed using Origin 2019b software. Results. The amount of ZEB1 in tumor tissue exceeds its concentration in conditionally normal (histologically unchanged) tissue by more than 5 times. Approximately the same ratio in the amount of ZEB1 between normal and tumor tissues is observed in samples from patients with РТС and metastasis. The concentration of transcription factors in metastasis exceeds the norm by almost 10 times. The amount of ZEB1 in goiter tissue does not differ from conditionally normal tissue of РТС patients but it was significantly different from tumor tissue of carcinoma. Conclusions. Our data indicate significant differences in ZEB1 concentration between conditionally normal and tumor thyroid tissues. The level of ZEB1 in metastasis was even higher. It should be emphasized that both in blood cells and plasma of metastatic PТC patients, the level of ZEB1 was significantly higher than in healthy people and individuals with PТC without metastasis. The latter circumstance can become the basis for the preoperative prognosis of the development of metastasis in PTC patients.
папілярна карцинома щитоподібної залози; метастазування; білок ZEB1; епітеліально-мезенхімальний перехід
papillary thyroid carcinoma; metastasis; ZEB1; epithelial-mesenchymal transition
Introduction
Materials and methods
Results
/17.jpg)
Discussion
Conclusions
- Nieto MA, Huang RY, Jackson RA. Thiery JP. EMT: 2016. Cell. 2016 Jun 30;166(1):21-45. doi: 10.1016/j.cell.2016.06.028.
- Zinych PP, Pushkarev VM, Levchuk NI, Tronko MD. ZEB1 as a marker of metastasis in thyroid carcinomas: review of literature and own data. Mìžnarodnij endokrinologìčnij žurnal. 2024;20(7):517-528. doi: 10.22141/2224-0721.20.7.2024.1451 (in Ukrainian).
- Welch DR, Hurst DR. Defining the Hallmarks of Metastasis. Cancer Res. 2019 Jun 15;79(12):3011-3027. doi: 10.1158/0008-5472.CAN-19-0458.
- Varun BR, Ramani P, Arya I, Palani J, Joseph AP. Epithelial-–mesenchymal transition in cancer stem cells: Therapeutic implications. J Oral Maxillofac Pathol. 2023;27:359-63. doi: 10.4103/jomfp.jomfp_308_22.
- Zinich PP, Pushkarev VM, Bolgov MYu, Guda BB, Pushka–rev VV. Molecular mechanisms of the formation of metastases. Markers of metastasis in thyroid carcinoma (review literary). Endokrynologia. 2020;25(3):227-242. doi: 10.31793/1680-1466.2020.25-3.227 (in Ukrainian).
- Kang E, Seo J, Yoon H, Cho S. The Post-Translational Regulation of Epithelial-Mesenchymal Transition-Inducing Transcription Factors in Cancer Metastasis. Int J Mol Sci. 2021 Mar 30;22(7):3591. doi: 10.3390/ijms22073591.
- Vatseba TS, Sokolova LK, Pushkarev VM, et al. Activation of the PI3K/Akt/mTOR/p70S6K1 signaling cascade in the mononuclear cells of peripheral blood: Association with insulin and insulin-like growth factor levels in the blood of cancer patients and diabetes. Cytology and Genetics. 2019;53(6):489-493. doi: 10.3103/S0095452719060112.
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72(1):248-254.
- Krebs AM, Mitschke J, Lasierra Losada M, et al. The EMT-activator Zeb1 is a key factor for cell plasticity and promotes metastasis in pancreatic cancer. Nat Cell Biol. 2017 May;19(5):518-529. doi: 10.1038/ncb3513.
- Stemmler MP, Eccles RL, Brabletz S, Brabletz T. Non-redundant functions of EMT transcription factors. Nat Cell Biol. 2019 Jan;21(1):102-112. doi: 10.1038/s41556-018-0196-y.
- Caramel J, Ligier M, Puisieux A. Pleiotropic Roles for ZEB1 in Cancer. Cancer Res. 2018 Jan 1;78(1):30-35. doi: 10.1158/0008-5472.CAN-17-2476.
- Drápela S, Bouchal J, Jolly MK, Culig Z, Souček K. ZEB1: A Critical Regulator of Cell Plasticity, DNA Damage Response, and Therapy Resistance. Front Mol Biosci. 2020 Mar 19;7:36. doi: 10.3389/fmolb.2020.00036.
- Lu J, Fei F, Wu C, Mei J, Xu J, Lu P. ZEB1: Catalyst of immune escape during tumor metastasis. Biomed Pharmacother. 2022 Sep;153:113490. doi: 10.1016/j.biopha.2022.113490.
- Celià-Terrassa T, Jolly MK. Cancer Stem Cells and Epithe–lial-to-Mesenchymal Transition in Cancer Metastasis. Cold Spring Harb Perspect Med. 2020 Jul 1;10(7):a036905. doi: 10.1101/cshperspect.a036905.
- Vasko V. Thyroid Cancer: Focus on Invasion and Metastasis Mechanisms, Therapeutic Target and Drug Treatment. Cancers (Basel). 2023 Sep 28;15(19):4762. doi: 10.3390/cancers15194762.
- Zhang J, Xu S. High aggressiveness of papillary thyroid cancer: from clinical evidence to regulatory cellular networks. Cell Death Discov. 2024;10:378. doi: 10.1038/s41420-024-02157-2.
- Guda BB, Pushkarev VV, Kovzun OI, Pushkarev VM, Tronko MD. PCNA expression as a marker of proliferation in benign and highly differentiated malignant tumors of the human thyroid gland (literature review and clinical case) Mìžnarodnij endokrinologìčnij žurnal. 2019;15(4):339-343. doi: 10.22141/2224-0721.15.4.2019.174822 (in Ukrainian).
- Garifulin OM, Filonenko VV, Bdzhola AV, et al. Expression of Ribosomal Protein S6 Kinase (S6K1) Isoforms in Different Types of Papillary Thyroid Carcinoma. Cytol Genet. 2023;57:305-311. doi: 10.3103/S0095452723040059.
- Zinych PP, Pushkarev VM, Levchuk NI, Shelkovoy EA, Bolgov MYu. Expression of tight junction protein ZO-1 in thyroid carcinomas. Endokrynologia. 2025;30(4):291-295. doi: 10.31793/1680-1466.2025.30-3.300.
- Kobrynska NYa, Pushkarev VM, Levchuk NI, Kovzun OI. Matrix metalloproteinase 2 level in blood plasma, thyroid tumors and metastases. Endokrynologia Polska. 2025;30(2):119-123. doi: 10.31793/1680-1466.2025.30-2.119.
- Morillo-Bernal J, Fernández LP, Santisteban P. FOXE1 regu–lates migration and invasion in thyroid cancer cells and targets ZEB1. Endocr Relat Cancer. 2020 Mar;27(3):137-151. doi: 10.1530/ERC-19-0156.
- Wang Q, Shang J, Zhang Y, Zhou Y, Tang L. MiR-451a restrains the growth and metastatic phenotypes of papillary thyroid carcinoma cells via inhibiting ZEB1. Biomed Pharmacother. 2020 Jul;127:109901. doi: 10.1016/j.biopha.2020.109901.
- Vedovatto S, Oliveira FD, Pereira LC, Scheffel TB, Becken–kamp LR, Bertoni APS, et al. CD73 mitigates ZEB1 expression in pa–pillary thyroid carcinoma. Cell Commun Signal. 2024 Feb 22;22(1):145. doi: 10.1186/s12964-024-01522-z.
- Liu R, Cao Z, Pan M, Wu M, Li X, Yuan H, Liu Z. A novel prognostic model for papillary thyroid cancer based on epi–thelial-mesenchymal transition-related genes. Cancer Med. 2022 Dec;11(23):4703-4720. doi: 10.1002/cam4.4836.
- Xiang Y, Wang W, Gu J, Shang J. Circular RNA VANGL1 Facilitates Migration and Invasion of Papillary Thyroid Cancer by Modulating the miR-194/ZEB1/EMT Axis. J Oncol. 2022 Mar 8;2022:4818651. doi: 10.1155/2022/4818651.
- Baldini E, Tuccilli C, Pironi D, Catania A, Tartaglia F, Di Matteo FM, et al. Expression and Clinical Utility of Transcription Factors Involved in Epithelial-Mesenchymal Transition during Thyroid Cancer Progression. J Clin Med. 2021 Sep 9;10(18):4076. doi: 10.3390/jcm10184076.