Международный эндокринологический журнал Том 22, №2, 2026
Вернуться к номеру
Епігенетичний вплив довгих некодуючих стеатогенних РНК на розвиток стеатотичної хвороби печінки, асоційованої з метаболічною дисфункцією
Авторы: A.E. Abaturov, A.O. Nikulina
Dnipro State Medical University, Dnipro, Ukraine
Рубрики: Эндокринология
Разделы: Справочник специалиста
Версия для печати
Актуальність. Стеатотична хвороба печінки, асоційована з метаболічною дисфункцією (metabolic dysfunction-associated steatotic liver disease — MASLD), є найпоширенішим хронічним захворюванням печінки в людей, що зумовлене зміною рівня експресії довгих некодуючіх РНК (long non-coding RNA — lncR). Метою цього огляду було надати короткий опис ролі стеатогенних lncR в епігенетичному впливі на розвиток MASLD шляхом аналізу даних сучасної наукової літератури. Матеріали та методи. Проведено аналіз 60 літературних джерел за останні п’ять років із баз даних MEDLINE; Embase; PreMedline In-Process & Other Non-Indexed Citations; The Cochrane Systematic Reviews Database, DARE, NHS EED and HTA databases; Web of Knowledge Science Citation Index; Web of Knowledge ISI Proceedings; CRD databases; BIOSIS, які були відібрані за ключовими словами: довгі некодуючі РНК; епігенетична регуляція; метаболічно асоційована жирова хвороба печінки; стеатотична хвороба печінки, асоційована з метаболічною дисфункцією; ожиріння. PROSPERO CRD420250652980. Результати. Посилення експресії стеатогенних lncR, як-от CCAT1, Gm10804, Gm15622, H19, HOTAIR, lncARSR, NEAT1, PVT1, SRA, Uc.372 та інші, є характерною ознакою розвитку й прогресування стеатозу печінки при MASLD. Розвиток стеатозу печінки при MASLD підтримується зниженням рівня експресії антистеатозних lncR, зокрема AC012668, FLRL2, Gm16551, lncHR1, B4GALT1-AS1/lncSHGL, MEG3, lncR MRAK052686 та інших. Висновки. Довгі некодуючі РНК мають безумовний патогенетичний вплив на основні механізми розвитку стеатозу печінки при MASLD, обумовлюючи поглинання ліпідів у гепатоцитах, посилюючи de novo ліпогенез, пригнічуючи β-окиснення жирних кислот та експорт ліпідів із гепатоцитів.
Background. Metabolic dysfunction-associated steatotic liver disease (MASLD) is the most common chronic liver disease in humans, which is caused by changes in the expression level of long non-coding RNAs (lncRs). The aim of this literature review is to present a brief description of the role of steatogenic lncRs in the epigenetic influence on the development of MASLD, analyzing the data of modern scientific literature. Materials and methods. An analysis of 60 literature sources over the past five years was conducted from the databases MEDLINE; Embase; PreMedline In-Process & Other Non-Indexed Citations; The Cochrane Systematic Reviews Database, DARE, NHS EED and HTA databases; Web of Knowledge Science Citation Index; Web of Knowledge ISI Proceedings; CRD databases; BIOSIS, which were selected using the following keywords: long non-coding RNAs, epigenetic regulation, metabolically associated fatty liver disease, metabolic dysfunction-associated steatotic liver disease obesity. PROSPERO CRD420250652980. Results. Increased expression of steatogenic lncRs, such as CCAT1, Gm10804, Gm15622, H19, HOTAIR, lncARSR, NEAT1, PVT1, SRA, Uc.372, is a characteristic feature of the development and progression of hepatic steatosis in MASLD. The development of hepatic steatosis in MASLD is supported by a decrease in the expression level of anti-steatotic lncRs, including AC012668, FLRL2, Gm16551, lncHR1, B4GALT1-AS1/lncSHGL, MEG3, lncR MRAK052686. Conclusions. Long noncoding RNAs have an unconditional pathogenetic influence on the main mechanisms of the development of hepatic steatosis in MASLD, promoting lipid uptake in hepatocytes, enhancing de novo lipogenesis, inhibiting β-oxidation of fatty acids and lipid export from hepatocytes.
епігенетична регуляція; довгі некодуючі стеатогенні РНК; стеатотична хвороба печінки, асоційована з метаболічною дисфункцією; ожиріння; огляд літератури
epigenetic regulation; long noncoding steatogenic RNAs; metabolic dysfunction-associated steatotic liver disease; obesity; literature review
Для ознакомления с полным содержанием статьи необходимо оформить подписку на журнал.
- NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet. 2017;390(10113):2627-42. doi: 10.1016/S0140-6736(17)32129-3.
- European Association for the Study of the Liver; European Association for the Study of Diabetes; European Association for the Study of Obesity. EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD): Executive Summary. Diabetologia. 2024 Nov;67(11):2375-2392. doi: 10.1007/s00125-024-06196-3.
- Faienza MF, Farella I, Khalil M, Portincasa P. Converging Pathways between Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD) and Diabetes in Children. Int J Mol Sci. 2024 Sep 14;25(18):9924. doi: 10.3390/ijms25189924.
- Panganiban J, Kehar M, Ibrahim SH, Hartmann P, Sood S, et al. Metabolic dysfunction-associated steatotic liver disease (MASLD) in children with obesity: An Obesity Medicine Association (OMA) and expert joint perspective 2025. Obes Pillars. 2025 Feb 1;14:100164. doi: 10.1016/j.obpill.2025.100164.
- Yang L, Li P, Yang W, Ruan X, Kiesewetter K, et al. Integrative Transcriptome Analyses of Metabolic Responses in Mice Define Pivotal LncRNA Metabolic Regulators. Cell Metab. 2016 Oct 11;24(4):627-639. doi: 10.1016/j.cmet.2016.08.019.
- DiStefano JK, Gerhard GS. Long Noncoding RNAs and Human Liver Disease. Ann Rev Pathol. 2022 Jan 24;17:1-21. doi: 10.1146/annurev-pathol-042320-115255.
- Alipoor B, Nikouei S, Rezaeinejad F, Malakooti-Dehkordi SN, Sabati Z, Ghasemi H. Long non-coding RNAs in metabolic disorders: pathogenetic relevance and potential biomarkers and therapeutic targets. J Endocrinol Invest. 2021 Oct;44(10):2015-2041. doi: 10.1007/s40618-021-01559-8.
- Abaturov OE, Nikulina AO. Characterization of anti-steatogenic long noncoding RNAs and their epigenetic influence on the development of metabolic fatty liver disease — a systematic review. Eur J Clin Exp Med. 2025;23(3):767-775. doi: 10.15584/ejcem.2025.3.16.
- Shi N, Sun K, Tang H, Mao J. The impact and role of identified long noncoding RNAs in nonalcoholic fatty liver disease: A narrative review. J Clin Lab Anal. 2023 Jun;37(11–12):e24943. doi: 10.1002/jcla.24943.
- Zeng Q, Liu CH, Wu D, Jiang W, Zhang N, Tang H. LncRNA and circRNA in Patients with Non-Alcoholic Fatty Liver Disease: A Systematic Review. Biomolecules. 2023 Mar 20;13(3):560. doi: 10.3390/biom13030560.
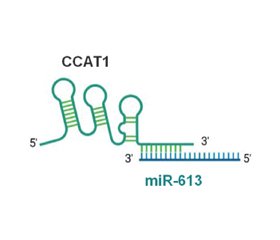
- Huang F, Liu H, Lei Z, Li Z, Zhang T, Yang M, et al. Long noncoding RNA CCAT1 inhibits miR-613 to promote nonalcoholic fatty liver disease via increasing LXRα transcription. J Cell Physiol. 2020 Dec;235(12):9819-9833. doi: 10.1002/jcp.29795.
- Leti F, DiStefano JK. Long Noncoding RNAs as Diagnostic and Therapeutic Targets in Type 2 Diabetes and Related Complications. Genes (Basel). 2017 Aug 22;8(8):207. doi: 10.3390/genes8080207.
- Tang Z, Li X, Zheng Y, Liu J, Liu C, Li X. The role of competing endogenous RNA network in the development of hepatocellular carcinoma: potential therapeutic targets. Front Cell Dev Biol. 2024 Jan 31;12:1341999. doi: 10.3389/fcell.2024.1341999.
- Liau XL, Salvamani S, Gunasekaran B, Chellappan DK, Rhodes A, et al. CCAT 1 — A Pivotal Oncogenic Long Non-Coding RNA in Colorectal Cancer. Br J Biomed Sci. 2023 Mar 21;80:11103. doi: 10.3389/bjbs.2023.11103.
- Liu Z, Chen Q, Hann SS. The functions and oncoge–nic roles of CCAT1 in human cancer. Biomed Pharmacother. 2019 Jul;115:108943. doi: 10.1016/j.biopha.2019.108943.
- Li T, Huang X, Yue Z, Meng L, Hu Y. Knockdown of long non-coding RNA Gm10804 suppresses disorders of hepatic glucose and lipid metabolism in diabetes with non-alcoholic fatty liver disease. Cell Biochem Funct. 2020 Oct;38(7):839-846. doi: 10.1002/cbf.3495.
- Ma M, Duan R, Shen L, Liu M, Ji Y, et al. The lncRNA Gm15622 stimulates SREBP-1c expression and hepatic lipid accumulation by sponging the miR-742-3p in mice. J Lipid Res. 2020 Jul;61(7):1052-1064. doi: 10.1194/jlr.RA120000664.
- Busscher D, Boon RA, Juni RP. The multifaceted actions of the lncRNA H19 in cardiovascular biology and diseases. Clin Sci (Lond). 2022 Aug 12;136(15):1157-1178. doi: 10.1042/CS20210994.
- Zhou J, Yang L, Zhong T, Mueller M, Men Y, et al. H19 lncRNA alters DNA methylation genome wide by regulating S-adenosylhomocysteine hydrolase. Nat Commun. 2015 Dec 21;6:10221. doi: 10.1038/ncomms10221.
- Zhang N, Geng T, Wang Z, Zhang R, Cao T, et al. Elevated hepatic expression of H19 long noncoding RNA contributes to diabetic hyperglycemia. JCI Insight. 2018 May 17;3(10):e120304. doi: 10.1172/jci.insight.120304.
- Wang H, Cao Y, Shu L, Zhu Y, Peng Q, et al. Long non-–coding RNA (lncRNA) H19 induces hepatic steatosis through activating MLXIPL and mTORC1 networks in hepatocytes. J Cell Mol Med. 2020 Jan;24(2):1399-1412. doi: 10.1111/jcmm.14818.
- Liu J, Tang T, Wang GD, Liu B. LncRNA-H19 promotes hepatic lipogenesis by directly regulating miR-130a/PPARγ axis in non-alcoholic fatty liver disease. Biosci Rep. 2019 Jul 15;39(7):BSR20181722. doi: 10.1042/BSR20181722.
- Liao J, Chen B, Zhu Z, Du C, Gao S, et al. Long nonco–ding RNA (lncRNA) H19: An essential developmental regulator with expanding roles in cancer, stem cell differentiation, and metabolic diseases. Genes Dis. 2023 Mar 24;10(4):1351-1366. doi: 10.1016/j.gendis.2023.02.008.
- Zhu J, Luo Z, Pan Y, Zheng W, Li W, et al. H19/miR-148a/USP4 axis facilitates liver fibrosis by enhancing TGF-β signaling in both hepatic stellate cells and hepatocytes. J Cell Physiol. 2019 Jun;234(6):9698-9710. doi: 10.1002/jcp.27656.
- Sun Y, Liu C, Guo X, Zhao J, Xiao A, et al. Identification of the c-Jun/H19/miR-19/JNK1 cascade during hepatic stellate cell activation. Clin Transl Med. 2023 Mar;13(3):e1106. doi: 10.1002/ctm2.1106.
- Lee GH, Peng C, Park SA, Hoang TH, Lee HY, et al. Citrus Peel Extract Ameliorates High-Fat Diet-Induced NAFLD via Activation of AMPK Signaling. Nutrients. 2020 Mar 1;12(3):673. doi: 10.3390/nu12030673.
- Guo B, Cheng Y, Yao L, Zhang J, Lu J, et al. LncRNA HOTAIR regulates the lipid accumulation in non-alcoholic fatty liver disease via miR-130b-3p/ROCK1 axis. Cell Signal. 2022 Feb;90:110190. doi: 10.1016/j.cellsig.2021.110190.
- Ferro A, Saccu G, Mattivi S, Gaido A, Herrera Sanchez MB, et al. Extracellular Vesicles as Delivery Vehicles for Non-Coding RNAs: Potential Biomarkers for Chronic Liver Diseases. Biomolecules. 2024 Feb 26;14(3):277. doi: 10.3390/biom14030277.
- Landry T, Shookster D, Huang H. Tissue-Specific Approaches Reveal Diverse Metabolic Functions of Rho-Kinase 1. Front Endocrinol (Lausanne). 2021 Feb 9;11:622581. doi: 10.3389/fendo.2020.622581.
- Zaky YA, Rashad MW, Zaater MA, El Kerdawy AM. Discovery of dual rho-associated protein kinase 1 (ROCK1)/apoptosis signal-regulating kinase 1 (ASK1) inhibitors as a novel approach for non-alcoholic steatohepatitis (NASH) treatment. BMC Chem. 2024 Jan 3;18(1):2. doi: 10.1186/s13065-023-01081-3.
- Zhang M, Chi X, Qu N, Wang C. Long noncoding RNA lncARSR promotes hepatic lipogenesis via Akt/SREBP-1c pathway and contributes to the pathogenesis of nonalcoholic steatohepatitis. Biochem Biophys Res Commun. 2018 Apr 30;499(1):66-70. doi: 10.1016/j.bbrc.2018.03.127.
- Chi Y, Gong Z, Xin H, Wang Z, Liu Z. Long noncoding RNA lncARSR promotes nonalcoholic fatty liver disease and hepatocellular carcinoma by promoting YAP1 and activating the IRS2/AKT pathway. J Transl Med. 2020 Mar 13;18(1):126. doi: 10.1186/s12967-020-02225-y.
- Li N, Li X, Ding Y, Liu X, Diggle K, et al. SREBP Regulation of Lipid Metabolism in Liver Disease, and Therapeutic Strategies. Biomedicines. 2023 Dec 12;11(12):3280. doi: 10.3390/biomedicines11123280.
- Huang J, Chen S, Cai D, Bian D, Wang F. Long noncoding RNA lncARSR promotes hepatic cholesterol biosynthesis via modulating Akt/SREBP-2/HMGCR pathway. Life Sci. 2018 Jun 15;203:48-53. doi: 10.1016/j.lfs.2018.04.028.
- Ji P, Diederichs S, Wang W, Böing S, Metzger R, et al. MALAT-1, a novel noncoding RNA, and thymosin beta4 predict meta–stasis and survival in early-stage non-small cell lung cancer. Oncogene. 2003 Sep 11;22(39):8031-41. doi: 10.1038/sj.onc.1206928.
- Tufail M. The MALAT1-breast cancer interplay: insights and implications. Expert Rev Mol Diagn. 2023 Jul-Dec;23(8):665-678. doi: 10.1080/14737159.2023.2233902.
- Piórkowska K, Zygmunt K, Hunter W, Wróblewska K. MALAT1: A Long Non-Coding RNA with Multiple Functions and Its Role in Processes Associated with Fat Deposition. Genes (Basel). 2024 Apr 10;15(4):479. doi: 10.3390/genes15040479.
- Yan C, Chen J, Chen N. Long noncoding RNA MALAT1 promotes hepatic steatosis and insulin resistance by increasing nu–clear SREBP-1c protein stability. Sci Rep. 2016 Mar 3;6:22640. doi: 10.1038/srep22640.
- Han J, Shen L, Zhan Z, Liu Y, Zhang C, et al. The long noncoding RNA MALAT1 modulates adipose loss in cancer-associated cachexia by suppressing adipogenesis through PPAR-γ. Nutr Metab (Lond). 2021 Mar 10;18(1):27. doi: 10.1186/s12986-021-00557-0.
- Xiang J, Deng YY, Liu HX, Pu Y. LncRNA MALAT1 Promotes PPARα/CD36-Mediated Hepatic Lipogenesis in Nonalcoholic Fatty Liver Disease by Modulating miR-206/ARNT Axis. Front Bioeng Biotechnol. 2022 Jun 13;10:858558. doi: 10.3389/fbioe.2022.858558.
- Chen X, Tan QQ, Tan XR, Li SJ, Zhang XX. Circ_0057558 promotes nonalcoholic fatty liver disease by regulating ROCK1/AMPK signaling through targeting miR-206. Cell Death Dis. 2021 Aug 26;12(9):809. doi: 10.1038/s41419-021-04090-z.
- Bougarne N, Weyers B, Desmet SJ, Deckers J, Ray DW, et al. Molecular Actions of PPARα in Lipid Metabolism and Inflammation. Endocr Rev. 2018 Oct 1;39(5):760-802. doi: 10.1210/er.2018-00064.
- Chen S, Sun S, Feng Y, Li X, Yin G, et al. Diosgenin attenua–tes nonalcoholic hepatic steatosis through the hepatic FXR-SHP-SREBP1C/PPARα/CD36 pathway. Eur J Pharmacol. 2023 Aug 5;952:175808. doi: 10.1016/j.ejphar.2023.175808.
- Wang Z, Li K, Huang W. Long non-coding RNA NEAT1-centric gene regulation. Cell Mol Life Sci. 2020 Oct;77(19):3769-3779. doi: 10.1007/s00018-020-03503-0.
- Jin SS, Lin CJ, Lin XF, Zheng JZ, Guan HQ. Silencing lncRNA NEAT1 reduces nonalcoholic fatty liver fat deposition by regu–lating the miR-139-5p/c-Jun/SREBP-1c pathway. Ann Hepatol. 2022 Mar-Apr;27(2):100584. doi: 10.1016/j.aohep.2021.100584.
- Sun Y, Song Y, Liu C, Geng J. LncRNA NEAT1-Micro–RNA-140 axis exacerbates nonalcoholic fatty liver through interrupting AMPK/SREBP-1 signaling. Biochem Biophys Res Commun. 2019 Aug 20;516(2):584-590. doi: 10.1016/j.bbrc.2019.06.104.
- Hu MJ, Long M, Dai RJ. Acetylation of H3K27 activated lncRNA NEAT1 and promoted hepatic lipid accumulation in non-alcoholic fatty liver disease via regulating miR-212-5p/GRIA3. Mol Cell Biochem. 2022 Jan;477(1):191-203. doi: 10.1007/s11010-021-04269-0.
- Guo Y, Yu J, Wang C, Li K, Liu B, et al. miR-212-5p suppresses lipid accumulation by targeting FAS and SCD1. J Mol Endocrinol. 2017 Oct;59(3):205-217. doi: 10.1530/JME-16-0179.
- Chen X, Tan XR, Li SJ, Zhang XX. LncRNA NEAT1 promotes hepatic lipid accumulation via regulating miR-146a-5p/ROCK1 in nonalcoholic fatty liver disease. Life Sci. 2019 Oct 15;235:116829. doi: 10.1016/j.lfs.2019.116829.
- Jin SS, Lin XF, Zheng JZ, Wang Q, Guan HQ. lncRNA NEAT1 regulates fibrosis and inflammatory response induced by nonalcoholic fatty liver by regulating miR-506/GLI3. Eur Cytokine Netw. 2019 Sep 1;30(3):98-106. doi: 10.1684/ecn.2019.0432.
- Wang X, Ma Y, Yang LY, Zhao D. MicroRNA-20a-5p Ameliorates Non-alcoholic Fatty Liver Disease via Inhibiting the Expression of CD36. Front Cell Dev Biol. 2020 Dec 3;8:596329. doi: 10.3389/fcell.2020.596329..
- Zhang H, Niu Q, Liang K, Li X, Jiang J, Bian C. Effect of LncPVT1/miR-20a-5p on Lipid Metabolism and Insulin Resistance in NAFLD. Diabetes Metab Syndr Obes. 2021 Nov 23;14:4599-4608. doi: 10.2147/DMSO.S338097.
- Sheng L, Ye L, Zhang D, Cawthorn WP, Xu B. New Insights Into the Long Non-coding RNA SRA: Physiological Functions and Mechanisms of Action. Front Med (Lausanne). 2018 Sep 6;5:244. doi: 10.3389/fmed.2018.00244.
- Shabgah AG, Norouzi F, Hedayati-Moghadam M, Soleimani D, Pahlavani N, Navashenaq JG. A comprehensive review of long non-coding RNAs in the pathogenesis and development of non-alcoholic fatty liver disease. Nutr Metab (Lond). 2021 Feb 23;18(1):22. doi: 10.1186/s12986-021-00552-5.
- Guo J, Fang W, Sun L, Lu Y, Dou L, et al. Ultraconserved element uc.372 drives hepatic lipid accumulation by suppressing miR-195/miR4668 maturation. Nat Commun. 2018 Feb 9;9(1):612. doi: 10.1038/s41467-018-03072-8.
- Chen X, Ma H, Gao Y, et al. Long non-coding RNA AC012668 suppresses non-alcoholic fatty liver disease by competing for microRNA miR-380-5p with lipoprotein-related protein LRP2. Bioengineered. 2021;12(1):6738-6747. doi: 10.1080/21655979.2021.1960463.
- Qian W, Liu Z, Zhang W, et al. Targeting steroid receptor RNA activator as a novel therapeutic strategy for myocardial hypertrophy. Biochem Pharmacol. 2025;240:117121. doi: 10.1016/j.bcp.2025.117121.