Журнал "Гастроэнтерология" Том 60, №1, 2026
Вернуться к номеру
Інтерлейкін-22 при гострому панкреатиті: коли цитопротекція стає ризиком
Авторы: Чуклін С.М., Чуклін С.С.
Медичний центр Святої Параскеви, м. Львів, Україна
Рубрики: Гастроэнтерология
Разделы: Клинические исследования
Версия для печати
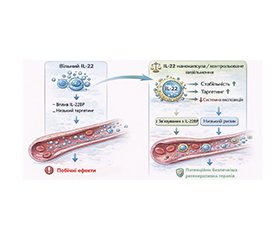
Актуальність. Гострий панкреатит (ГП) — тяжкий запальний стан, що характеризується передчасною активацією травних ферментів, ушкодженням ацинарних клітин і формуванням системних ускладнень. Інтерлейкін-22 (IL-22), представник родини IL-10, привертає дедалі більшу увагу як медіатор на перетині імунної регуляції та епітеліального захисту. Накопичені експериментальні дані свідчать про контекстуальну залежність його ефектів: залежно від стадії ГП та характеристик імунного мікросередовища IL-22 може реалізовувати як протективні, так і потенційно патогенні властивості. Матеріали та методи. У цьому огляді узагальнено результати експериментальних і клінічних досліджень, присвячених вивченню ролі IL-22 при ГП. Проаналізовано дані мишачих моделей, експериментів in vitro та клінічних когортних спостережень із фокусом на сигнальних механізмах, тканинозахисних і ушкоджувальних ефектах, а також трансляційному потенціалі IL-22 як терапевтичної мішені та біомаркера. Результати. IL-22 забезпечує цитопротекцію ацинарних клітин через активацію сигнальних шляхів STAT3 і AKT/mTOR, обмежує надмірну автофагію та індукує експресію антиапоптичних білків (Bcl-2, Bcl-xL). Цитокін також підтримує цілісність кишкового бар’єра шляхом індукції Reg3β/γ, зменшує ураження легень завдяки стримуванню нейтрофільного запалення та демонструє гепатопротекторні й ренопротекторні властивості. У пацієнтів із тяжким ГП підвищені сироваткові рівні IL-22 асоціюються з розвитком шлунково-кишкової недостатності та системних ускладнень, що підкреслює його потенційну прогностичну цінність як біомаркера. Водночас IL-22 характеризується функціональною амбівалентністю: за певних умов при легкому ГП він може посилювати некротичні зміни, тоді як тривала активація IL-22-залежних сигнальних каскадів асоціюється з фіброгенезом і канцерогенезом. Критичним компонентом підтримання гомеостазу є негативна регуляція через білок-зв’язувач IL-22 (IL-22BP). Перспективним напрямом доклінічних досліджень залишаються нанотерапевтичні підходи, спрямовані на підвищення стабільності та ефективності доставки IL-22. Висновки. IL-22 розглядається як перспективний терапевтичний кандидат і потенційний біомаркер при ГП. Рекомбінантний IL-22 та сучасні системи доставки можуть знижувати ризик ускладнень при тяжкому перебігу захворювання, тоді як визначення IL-22 у крові здатне поліпшити прогностичну стратифікацію. Водночас залишаються невирішеними питання безпеки, оптимального терапевтичного вікна та довгострокових ризиків. Для інтеграції IL-22 у підходи персоналізованої медицини при ГП потрібні масштабні, методологічно коректно спроєктовані клінічні випробування.
Background. Acute pancreatitis (AP) is a severe inflammatory disorder characterized by premature activation of digestive enzymes, acinar cell injury, and systemic complications. Interleukin-22 (IL-22), a cytokine of the IL-10 family, has emerged as a key mediator linking immune regulation with epithelial protection. Experimental data suggest that IL-22 exerts both protective and pathogenic effects depending on disease stage and immune context. Materials and methods. This review synthesizes findings from experimental and clinical studies on IL-22 in AP. Literature from murine models, in vitro studies, and patient cohorts was analyzed with a focus on signaling mechanisms, tissue-protective versus pathogenic roles, and translational potential as a therapeutic target and biomarker. Results. IL-22 protects acinar cells via STAT3 and AKT/mTOR signaling, inhibits excessive autophagy, and induces anti-apoptotic proteins (Bcl-2, Bcl-xL). It enhances intestinal barrier function through Reg3β/γ induction, mitigates lung injury by restraining neutrophilic inflammation, and shows potential hepatoprotective and renoprotective effects. Elevated serum IL-22 levels in patients with severe AP correlate with gastrointestinal failure and systemic complications, suggesting its potential prognostic value as a biomarker. However, IL-22 displays functional duality: in mild AP, it may worsen necrosis, while chronic activation of IL-22-dependent signaling cascades has been associated with fibrosis and tumorigenesis. Negative regulation through IL-22 binding protein is critical component in maintaining homeostasis. Preclinical nanotherapeutic approaches have improved IL-22 delivery and stability. Conclusions. IL-22 represents both a promising therapeutic target and a potential biomarker in AP. Recombinant IL-22 and advanced delivery platforms could reduce complications in severe disease, while IL-22 measurement may refine prognostic stratification. Nevertheless, challenges remain regarding safety, therapeutic windows, and long-term risks. Large-scale clinical studies are needed to establish IL-22 as a cornerstone of precision medicine in AP.
гострий панкреатит; інтерлейкін-22; цитокіни; передача сигналів; біомаркери
acute pancreatitis; interleukin-22; cytokines; signal transduction; biomarkers
Для ознакомления с полным содержанием статьи необходимо оформить подписку на журнал.
- Boxhoorn L, Voermans RP, Bouwense SA, Bruno MJ, Verdonk RC, Boermeester MA, et al. Acute pancreatitis. Lancet. 2020 Sep 5;396(10252):726-734. doi: 10.1016/S0140-6736(20)31310-6. PMID: 32891214.
- Yu L, Xie F, Luo L, Lei Y, Huang X, Yang X, et al. Clinical characteristics and risk factors of organ failure and death in necrotizing pancreatitis. BMC Gastroenterol. 2023 Jan 19;23(1):19. doi: 10.1186/s12876-023-02651-4. PMID: 36658497.
- Li T, Qin C, Zhao B, Li Z, Zhao Y, Lin C, et al. Global and regional burden of pancreatitis: epidemiological trends, risk factors, and projections to 2050 from the global burden of disease study 2021. BMC Gastroenterol. 2024 Nov 8;24(1):398. doi: 10.1186/s12876-024-03481-8. PMID: 39511469.
- Weiss FU, Laemmerhirt F, Lerch MM. Etiology and risk factors of acute and chronic pancreatitis. Visc Med. 2019 Apr;35(2):73-81. doi: 10.1159/000499138. Epub 2019 Mar 13. PMID: 31192240.
- Yuan S, Giovannucci EL, Larsson SC. Gallstone disease, diabetes, calcium, triglycerides, smoking and alcohol consumption and pancreatitis risk: Mendelian randomization study. NPJ Genom Med. 2021 Mar 29;6(1):27. doi: 10.1038/s41525-021-00189-6. PMID: 33782414.
- Chen X, Zhong R, Hu B. Mitochondrial dysfunction in the pathogenesis of acute pancreatitis. Hepatobiliary Pancreat Dis Int. 2025 Feb;24(1):76-83. doi: 10.1016/j.hbpd.2023.12.008. Epub 2023 Dec 30. PMID: 38212158.
- Zheng Z, Ding YX, Qu YX, Cao F, Li F. A narrative review of acute pancreatitis and its diagnosis, pathogenetic mechanism, and management. Ann Transl Med. 2021 Jan;9(1):69. doi: 10.21037/atm-20-4802. PMID: 33553362.
- Coman LI, Balaban DV, Dumbravă BF, Păunescu H, Marin RC, Costescu M, et al. Targeting oxidative stress in acute pancreatitis: a critical review of antioxidant strategies. Nutrients. 2025 Jul 22;17(15):2390. doi: 10.3390/nu17152390. PMID: 40805976.
- Sternby H, Hartman H, Thorlacius H, Regnér S. The initial course of IL-1β, IL-6, IL-8, IL-10, IL-12, IFN-γ and TNF-α with regard to severity grade in acute pancreatitis. Biomolecules. 2021 Apr 17;11(4):591. doi: 10.3390/biom11040591. PMID: 33920566.
- Rafaqat S, Patoulias D, Behnoush AH, Sharif S, Klisic A. Interleukins: pathophysiological role in acute pancreatitis. Arch Med Sci. 2024 Jan 31;20(1):138-156. doi: 10.5114/aoms/178183. PMID: 38414463.
- Liu Q, Zhu X, Guo S. From pancreas to lungs: the role of immune cells in severe acute pancreatitis and acute lung injury. Immun Inflamm Dis. 2024 Jul;12(7):e1351. doi: 10.1002/iid3.1351. PMID: 39023414.
- Manohar M, Jones EK, Rubin SJS, Subrahmanyam PB, Swaminathan G, Mikhail D, et al. Novel circulating and tissue monocytes as well as macrophages in pancreatitis and recovery. Gastroenterology. 2021 Dec;161(6):2014-2029.e14. doi: 10.1053/j.gastro.2021.08.033. Epub 2021 Aug 25. PMID: 34450180.
- Xu Q, Fu X, Xiu Z, Yang H, Men X, Liu M, et al. Interleukin 22 alleviates arginine induced pancreatic acinar cell injury via the regulation of intracellular vesicle transport system: evidence from proteomic analysis. Exp Ther Med. 2023 Dec;26(6):578. doi: 10.3892/etm.2023.12277. Epub 2023 Oct 26. PMID: 38023358.
- Dumoutier L, Van Roost E, Colau D, Renauld JC. Human interleukin-10-related T cell-derived inducible factor: molecular cloning and functional characterization as an hepatocyte-stimulating factor. Proc Natl Acad Sci USA. 2000 Aug 29;97(18):10144-9. doi: 10.1073/pnas.170291697. PMID: 10954742.
- Yang H, Cao R, Zhou F, Wang B, Xu Q, Li R, et al. The role of interleukin-22 in severe acute pancreatitis. Mol Med. 2024 May 15;30(1):60. doi: 10.1186/s10020-024-00826-7. PMID: 38750415.
- Tsomidis I, Voumvouraki A, Kouroumalis E. The pathoge–nesis of pancreatitis and the role of autophagy. Gastroenterol Insights. 2024;15(2):303-341. doi: 10.3390/gastroent15020022.
- Dong H, Zhu F, Jin S, Tian J. Interleukin-22 regulates gastric cancer cell proliferation through regulation of the JNK signaling pathway. Exp Ther Med. 2020 Jul;20(1):205-210. doi: 10.3892/etm.2020.8707. Epub 2020 Apr 30. PMID: 32536992.
- Fu X, Xiu Z, Xu Q, Yue R, Xu H. Interleukin-22 alleviates caerulein-induced acute pancreatitis by activating AKT/mTOR pathway. Dig Dis Sci. 2024 May;69(5):1691-1700. doi: 10.1007/s10620-024-08360-6. Epub 2024 Mar 11. PMID: 38466463.
- Bai J, Bai J, Yang M. Interleukin-22 attenuates acute pan–creatitis-associated intestinal mucosa injury in mice via STAT3 activation. Gut Liver. 2021 Sep 15;15(5):771-781. doi: 10.5009/gnl20210. PMID: 33495423.
- Qiao YY, Liu XQ, Xu CQ, Zhang Z, Xu HW. Interleukin-22 ameliorates acute severe pancreatitis-associated lung injury in mice. World J Gastroenterol. 2016 Jun 7;22(21):5023-32. doi: 10.3748/wjg.v22.i21.5023. PMID: 27275094.
- Cineus R, Luo Y, Saliutina M, Manna S, Cancino CA, Vela–sco Blázquez L, et al.; TRR241 IBDome Consortium. The IL-22-oncostatin M axis promotes intestinal inflammation and tumorigenesis. Nat Immunol. 2025 Jun;26(6):837-853. doi: 10.1038/s41590-025-02149-z. Epub 2025 May 30. PMID: 40447860.
- Zhao N, Liu C, Li N, Zhou S, Guo Y, Yang S, et al. Role of interleukin-22 in ulcerative colitis. Biomed Pharmacother. 2023 Mar;159:114273. doi: 10.1016/j.biopha.2023.114273. Epub 2023 Jan 23. PMID: 36696801.
- Sajiir H, Ramm GA, Macdonald GA, McGuckin MA, Prins JB, Hasnain SZ. Harnessing IL-22 for metabolic health: promise and pitfalls. Trends Mol Med. 2025 Jun;31(6):574-584. doi: 10.1016/j.molmed.2024.10.016. Epub 2024 Nov 21. PMID: 39578121.
- Dean LS, Threatt AN, Jones K, Oyewole EO, Pauly M, Wahl M, et al. I don’t know about you, but I’m feeling IL-22. Cytokine Growth Factor Rev. 2024 Dec;80:1-11. doi: 10.1016/j.cytogfr.2024.11.001. Epub 2024 Nov 8. PMID: 39537498.
- Saxton RA, Henneberg LT, Calafiore M, Su L, Jude KM, Hanash AM, et al. The tissue protective functions of interleukin-22 can be decoupled from pro-inflammatory actions through structure-based design. Immunity. 2021 Apr 13;54(4):660-672.e9. doi: 10.1016/j.immuni.2021.03.008. PMID: 33852830.
- Mihi B, Gong Q, Nolan LS, Gale SE, Goree M, Hu E, et al. Interleukin-22 signaling attenuates necrotizing enterocolitis by promoting epithelial cell regeneration. Cell Rep Med. 2021 Jun 15;2(6):100320. doi: 10.1016/j.xcrm.2021.100320. PMID: 34195684.
- Li J, Wu Z, Wu Y, Hu X, Yang J, Zhu D, et al. IL-22, a vital cytokine in autoimmune diseases. Clin Exp Immunol. 2024;218(3):242-263. doi: 10.1093/cei/uxae035. PMID: 38651179.
- Lindahl H, Olsson T. Interleukin-22 influences the Th1/Th17 axis. Front Immunol. 2021 Feb 22;12:618110. doi: 10.3389/fimmu.2021.618110. PMID: 33692792.
- Jin M, Zhang H, Wu M, Wang Z, Chen X, Guo M, et al. Colonic interleukin-22 protects intestinal mucosal barrier and microbiota abundance in severe acute pancreatitis. FASEB J. 2022 Mar;36(3):e22174. doi: 10.1096/fj.202101371R. PMID: 35137988.
- Mar JS, Ota N, Pokorzynski ND, Peng Y, Jaochico A, Sangaraju D, et al. IL-22 alters gut microbiota composition and function to increase aryl hydrocarbon receptor activity in mice and humans. Microbiome. 2023 Mar 9;11(1):47. doi: 10.1186/s40168-023-01486-1. PMID: 36894983.
- Didriksen BJ, Eshleman EM, Alenghat T. Epithelial regulation of microbiota-immune cell dynamics. Mucosal Immunol. 2024 Apr;17(2):303-313. doi: 10.1016/j.mucimm.2024.02.008. Epub 2024 Feb 28. PMID: 38428738.
- Yu Y, Qiao M, Liu J, Guo Y, Sun Y. The reciprocal regulation between autophagy and IL-22: implications for immunity and therapy. Clin Exp Med. 2025;25(1):187. doi: 10.1007/s10238-025-01695-y. PMID: 40465058.
- Aggarwal S, Xie MH, Maruoka M, Foster J, Gurney AL. Acinar cells of the pancreas are a target of interleukin-22. J Interferon Cytokine Res. 2001 Dec;21(12):1047-53. doi: 10.1089/107999001317205178. PMID: 11798462.
- Xuan X, Zhang L, Tian C, Wu T, Ye H, Cao J, et al. Interleukin-22 and connective tissue diseases: emerging role in pathogenesis and therapy. Cell Biosci. 2021 Jan 6;11(1):2. doi: 10.1186/s13578-020-00504-1. PMID: 33407883.
- Zenewicz LA. IL-22 binding protein (IL-22BP) in the regulation of IL-22 biology. Front Immunol. 2021 Nov 16;12:766586. doi: 10.3389/fimmu.2021.766586. PMID: 34868019.
- Seth P, Dubey S. IL-22 as a target for therapeutic intervention: current knowledge on its role in various diseases. Cytokine. 2023 Sep;169:156293. doi: 10.1016/j.cyto.2023.156293. Epub 2023 Jul 11. PMID: 37441942.
- Arshad T, Mansur F, Palek R, Manzoor S, Liska V. A double edged sword role of interleukin-22 in wound healing and tissue regeneration. Front Immunol. 2020 Sep 17;11:2148. doi: 10.3389/fimmu.2020.02148. PMID: 33042126.
- Fachi JL, Di Luccia B, Gilfillan S, Chang HW, Song C, Cheng J, et al. Deficiency of IL-22-binding protein enhances the abi–lity of the gut microbiota to protect against enteric pathogens. Proc Natl Acad Sci USA. 2024 May 7;121(19):e2321836121. doi: 10.1073/pnas.2321836121. Epub 2024 Apr 30. PMID: 38687788.
- Yan J, Yu J, Liu K, Liu Y, Mao C, Gao W. The pathogenic roles of IL-22 in colitis: its transcription regulation by musculin in T helper subsets and innate lymphoid cells. Front Immunol. 2021 Dec 21;12:758730. doi: 10.3389/fimmu.2021.758730. PMID: 34992594.
- Shohan M, Dehghani R, Khodadadi A, Dehnavi S, Ahmadi R, Joudaki N, et al. Interleukin-22 and intestinal homeostasis: protective or destructive? IUBMB Life. 2020 Aug;72(8):1585-1602. doi: 10.1002/iub.2295. Epub 2020 May 4. PMID: 32365282.
- Shin JH, Bozadjieva-Kramer N, Seeley RJ. Reg3γ: current understanding and future therapeutic opportunities in metabolic di–sease. Exp Mol Med. 2023 Aug;55(8):1672-1677. doi: 10.1038/s12276-023-01054-5. Epub 2023 Aug 1. PMID: 37524871.
- Luo M, Jin T, Fang Y, Chen F, Zhu L, Bai J, et al. Signaling pathways involved in acute pancreatitis. J Inflamm Res. 2025 Feb 17;18:2287-2303. doi: 10.2147/JIR.S485804. PMID: 40230438.
- Chakraborty D, Coslo DM, Murray IA, Vijay A, Patterson AD, Perdew GH. Immune cell-intrinsic Ah receptor facilitates the expression of antimicrobial REG3G in the small intestine. FASEB J. 2024 Feb 29;38(4):e23471. doi: 10.1096/fj.202302319R. PMID: 38358358.
- Ruan HX, Qin XN, Huang W, Lin L. IL-22 activates the PI3K-AKT pathway to promote colorectal cancer cell proliferation and metastasis. Discov Oncol. 2024 Jul 29;15(1):317. doi: 10.1007/s12672-024-01169-9. PMID: 39073546.
- Ma Q, Luan J, Bai Y, Xu C, Liu F, Chen B, et al. Interleukin-22 in renal protection and its pathological role in kidney di–seases. Front Immunol. 2022 Mar 31;13:851818. doi: 10.3389/fimmu.2022.851818. PMID: 35432360.
- Mahapatro M, Erkert L, Becker C. Cytokine-mediated crosstalk between immune cells and epithelial cells in the gut. Cells. 2021 Jan 9;10(1):111. doi: 10.3390/cells10010111. PMID: 33435303.
- Di Tommaso N, Gasbarrini A, Ponziani FR. Intestinal barrier in human health and disease. Int J Environ Res Public Health. 2021 Dec 6;18(23):12836. doi: 10.3390/ijerph182312836. PMID: 34886561.
- Xing Z, Wu Y, Liu N. IL-22 alleviates the fibrosis of hepatic stellate cells via the inactivation of NLRP3 inflammasome signaling. Exp Ther Med. 2021 Oct;22(4):1088. doi: 10.3892/etm.2021.10522. Epub 2021 Jul 30. PMID: 34447480.
- Sun L, Spiteri AG, Griffith BD, Zhang Y, Di Magliano MP, Olivei AC, et al. IL-22BP modulates injury in acute pancreatitis but delays tissue recovery in chronic pancreatitis. Cell Mol Gastroente–rol Hepatol. 2025;19(8):101520. doi: 10.1016/j.jcmgh.2025.101520. Epub 2025 Apr 22. PMID: 40274099.
- Zai W, Chen W, Liu H, Ju D. Therapeutic opportunities of IL-22 in non-alcoholic fatty liver disease: from molecular mechanisms to clinical applications. Biomedicines. 2021 Dec 14;9(12):1912. doi: 10.3390/biomedicines9121912. PMID: 34944732.
- Xu L, Cao P, Wang J, Zhang P, Hu S, Cheng C, et al. IL-22: a key inflammatory mediator as a biomarker and potential therapeutic target for lung cancer. Heliyon. 2024 Aug 10;10(17):e35901. doi: 10.1016/j.heliyon.2024.e35901. PMID: 39263114.
- Ding F, Liu G, Gao F, Zheng Z, Hong Y, Chen Y, et al. Adropin attenuates pancreatitis associated lung injury through PPARγ phosphorylation related macrophage polarization. Int J Mol Med. 2023 Oct;52(4):95. doi: 10.3892/ijmm.2023.5298. Epub 2023 Sep 1. PMID: 37654184.
- Huai JP, Sun XC, Chen MJ, Jin Y, Ye XH, Wu JS, et al. Melatonin attenuates acute pancreatitis-associated lung injury in rats by modulating interleukin 22. World J Gastroenterol. 2012 Sep 28;18(36):5122-8. doi: 10.3748/wjg.v18.i36.5122. PMID: 23049224.
- Kumar A, Patel VS, Harding JN, You D, Cormier SA. Exposure to combustion derived particulate matter exacerbates influenza infection in neonatal mice by inhibiting IL22 production. Part Fibre Toxicol. 2021 Dec 14;18(1):43. doi: 10.1186/s12989-021-00438-7. PMID: 34906172.
- Alcorn JF. IL-22 plays a critical role in maintaining epithelial integrity during pulmonary infection. Front Immunol. 2020 Jun 9;11:1160. doi: 10.3389/fimmu.2020.01160. PMID: 32582219.
- Zhou YD, Komnick MR, Sepulveda F, Liu G, Nieves-Ortiz E, Meador K, et al. Inducible, but not constitutive, pancreatic REG/Reg isoforms are regulated by intestinal microbiota and pancreatic diseases. Mucosal Immunol. 2025 Aug;18(4):918-936. doi: 10.1016/j.mucimm.2025.05.003. Epub 2025 May 19. PMID: 40398680.
- Li F, Wang Z, Cao Y, Pei B, Luo X, Liu J, et al. Intestinal mucosal immune barrier: a powerful firewall against severe acute pancreatitis-associated acute lung injury via the gut-lung axis. J Inflamm Res. 2024 Apr 10;17:2173-2193. doi: 10.2147/JIR.S448819. PMID: 38617383.
- Mo R, Lai R, Lu J, Zhuang Y, Zhou T, Jiang S, et al. Enhanced autophagy contributes to protective effects of IL-22 against acetaminophen-induced liver injury. Theranostics. 2018;8(15):4170-4180. doi: 10.7150/thno.25798. PMID: 30128045.
- Shao L, Xiong X, Zhang Y, Miao H, Ren Y, Tang X, et al. IL-22 ameliorates LPS-induced acute liver injury by autophagy activation through ATF4-ATG7 signaling. Cell Death Dis. 2020 Nov 11;11(11):970. doi: 10.1038/s41419-020-03176-4. PMID: 33177520.
- Jiang Z, Li W, Yu S, Wang X, Jiang H, Bai C, et al. IL-22 relieves hepatic ischemia-reperfusion injury by inhibiting mitochondrial apoptosis based on the activation of STAT3. Int J Biochem Cell Biol. 2024 Jan;166:106503. doi: 10.1016/j.biocel.2023.106503. Epub 2023 Nov 28. PMID: 38036287.
- Bai Y, Wu H, Zhang J, Zhang S, Zhang Z, Wang H, et al. IL-22 protects against biliary ischemia-reperfusion injury after li–ver transplantation via activating STAT3 and reducing apoptosis and oxidative stress levels in vitro and in vivo. Oxid Med Cell Longev. 2022 May 10;2022:9635075. doi: 10.1155/2022/9635075. PMID: 35592531.
- Xiang X, Feng D, Hwang S, Ren T, Wang X, Trojnar E, et al. Interleukin-22 ameliorates acute-on-chronic liver failure by reprogramming impaired regeneration pathways in mice. J Hepatol. 2020 Apr;72(4):736-745. doi: 10.1016/j.jhep.2019.11.013. Epub 2019 Nov 29. PMID: 31786256.
- Arab JP, Sehrawat TS, Simonetto DA, Verma VK, Feng D, Tang T, et al. An open-label, dose-escalation study to assess the safety and efficacy of IL-22 agonist F-652 in patients with alcohol-associated hepatitis. Hepatology. 2020 Aug;72(2):441-453. doi: 10.1002/hep.31046. Epub 2020 Apr 27. PMID: 31774566.
- Chen J, Sun S, Li H, Cai X, Wan C. IL-22 signaling promotes sorafenib resistance in hepatocellular carcinoma via STAT3/CD155 signaling axis. Front Immunol. 2024 Mar 25;15:1373321. doi: 10.3389/fimmu.2024.1373321. PMID: 38596684.
- Chen W, Shen Y, Fan J, Zeng X, Zhang X, Luan J, et al. IL-22-mediated renal metabolic reprogramming via PFKFB3 to treat kidney injury. Clin Transl Med. 2021 Feb;11(2):e324. doi: 10.1002/ctm2.324. PMID: 33634980.
- Shen Y, Jin X, Chen W, Gao C, Bian Q, Fan J, et al. Interleukin-22 ameliorated acetaminophen-induced kidney injury by inhibiting mitochondrial dysfunction and inflammatory responses. Appl Microbiol Biotechnol. 2020 Jul;104(13):5889-5898. doi: 10.1007/s00253-020-10638-4. Epub 2020 May 1. PMID: 32356198.
- Gu Y, Shen Y, Chen W, He H, Ma Y, Mei X, et al. Protective effects of interleukin-22 on oxalate-induced crystalline renal injury via alleviating mitochondrial damage and inflammatory response. Appl Microbiol Biotechnol. 2022 Apr;106(7):2637-2649. doi: 10.1007/s00253-022-11876-4. Epub 2022 Mar 16. PMID: 35294590.
- Taguchi K, Sugahara S, Elias BC, Pabla NS, Canaud G, Brooks CR. IL-22 is secreted by proximal tubule cells and regulates DNA damage response and cell death in acute kidney injury. Kidney Int. 2024 Jan;105(1):99-114. doi: 10.1016/j.kint.2023.09.020. Epub 2023 Dec 4. PMID: 38054920.
- Feng D, Park O, Radaeva S, Wang H, Yin S, Kong X, et al. Interleukin-22 ameliorates cerulein-induced pancreatitis in mice by inhibiting the autophagic pathway. Int J Biol Sci. 2012;8(2):249-257. doi: 10.7150/ijbs.3967. Epub 2012 Jan 6. PMID: 22253568.
- Huan C, Kim D, Ou P, Alfonso A, Stanek A. Mechanisms of interleukin-22’s beneficial effects in acute pancreatitis. World J Gastrointest Pathophysiol. 2016 Feb 15;7(1):108-116. doi: 10.4291/wjgp.v7.i1.108. PMID: 26909233.
- Glaubitz J, Asgarbeik S, Lange R, Mazloum H, Elsheikh H, Weiss FU, et al. Immune response mechanisms in acute and chronic pancreatitis: strategies for therapeutic intervention. Front Immunol. 2023 Oct 10;14:1279539. doi: 10.3389/fimmu.2023.1279539. PMID: 37881430.
- Vasseur P, Devaure I, Sellier J, Delwail A, Chagneau-Derrode C, Charier F, et al. High plasma levels of the pro-inflammatory cytokine IL-22 and the anti-inflammatory cytokines IL-10 and IL-1ra in acute pancreatitis. Pancreatology. 2014 Nov-Dec;14(6):465-469. doi: 10.1016/j.pan.2014.08.005. Epub 2014 Sep 6. PMID: 25240697.
- Protopsaltis NJ, Liang W, Nudleman E, Ferrara N. Interleukin-22 promotes tumor angiogenesis. Angiogenesis. 2019;22(3):311-323. doi: 10.1007/s10456-018-9658-x.
- Guendel F, Kofoed-Branzk M, Gronke K, Tizian C, Witkowski M, Cheng HW, et al. Group 3 innate lymphoid cells program a distinct subset of IL-22BP-producing dendritic cells demarcating solitary intestinal lymphoid tissues. Immunity. 2020 Nov 17;53(5):1015-1032.e8. doi: 10.1016/j.immuni.2020.10.012. PMID: 33207209.
- Stojanovic B, Jovanovic IP, Stojanovic MD, Jovanovic M, Vekic B, Milosevic B, et al. The emerging roles of the adaptive immune response in acute pancreatitis. Cells. 2023 May 29;12(11):1495. doi: 10.3390/cells12111495. PMID: 37296616.
- Mititelu A, Grama A, Colceriu MC, Pop TL. Overview of the cellular and immune mechanisms involved in acute pancreatitis: in search of new prognosis biomarkers. Expert Rev Mol Med. 2025 Jan 6;27:e9. doi: 10.1017/erm.2024.40. PMID: 39757373.
- Ding L, Yang Y, Li H, Wang H, Gao P. Circulating lymphocyte subsets induce secondary infection in acute pancreatitis. Front Cell Infect Microbiol. 2020 Mar 31;10:128. doi: 10.3389/fcimb.2020.00128. PMID: 32296650.
- Xue J, Nguyen DT, Habtezion A. Aryl hydrocarbon receptor regulates pancreatic IL-22 production and protects mice from acute pancreatitis. Gastroenterology. 2012 Dec;143(6):1670-1680. doi: 10.1053/j.gastro.2012.08.051. Epub 2012 Sep 27. PMID: 23022954.
- Ouyang W, O’Garra A. IL-10 family cytokines IL-10 and IL-22: from basic science to clinical translation. Immunity. 2019 Apr 16;50(4):871-891. doi: 10.1016/j.immuni.2019.03.020. PMID: 30995504.
- Soliman GA, Alamri MA, Abdel-Rahman RF, Elbaset MA, Ogaly HA, Abdel-Kader MS, et al. Tephrosia purpurea, with (-)-pseudosemiglabrin as the major constituent, alleviates severe acute pancreatitis-media–ted acute lung injury by modulating HMGB1 and IL-22. Int J Mol Sci. 2025 Mar 13;26(6):2572. doi: 10.3390/ijms26062572. PMID: 40141214.
- Alabbas SY, Begun J, Florin TH, Oancea I. The role of IL-22 in the resolution of sterile and nonsterile inflammation. Clin Transl Immunology. 2018 Apr 18;7(4):e1017. doi: 10.1002/cti2.1017. PMID: 29713472.
- Al-Hariri MT, Eldin TG, Hashim T, Chathoth S, Alswied A. Propolis modulates inflammatory mediators and improves histopatho–logy in male rats with L-arginine-induced acute pancreatitis. Sultan Qaboos Univ Med J. 2019 May;19(2):e103-e107. doi: 10.18295/squmj.2019.19.02.004. Epub 2019 Sep 8. PMID: 31538007.
- Lin J, Wei Y, Gu X, Liu M, Wang M, Zhou R, et al. Nano–therapeutics-mediated restoration of pancreatic homeostasis and intestinal barrier for the treatment of severe acute pancreatitis. J Control Release. 2025 Jan 10;377:93-105. doi: 10.1016/j.jconrel.2024.11.022. Epub 2024 Nov 17. PMID: 39542256.
- Sabat R, Ouyang W, Wolk K. Therapeutic opportunities of the IL-22-IL-22R1 system. Nat Rev Drug Discov. 2014 Jan;13(1):21-38. doi: 10.1038/nrd4176. PMID: 24378801.
- Orbelian L, Trembach N, Durleshter V. The role of inflammatory and hemostatic markers in the prediction of severe acute pancreatitis: an observational cohort study. Recent Adv Inflamm Allergy Drug Discov. 2024 Dec 17. doi: 10.2174/0127722708356543241209060544. Online ahead of print. PMID: 39694959.
- Fu X, Xiu Z, Xu H. Interleukin-22 and acute pancreatitis: a review. Medicine (Baltimore). 2023 Nov 3;102(44):e35695. doi: 10.1097/MD.0000000000035695. PMID: 37933011.
- Wagner F, Mansfield JC, Lekkerkerker AN, Wang Y, Keir M, Dash A, et al. Dose escalation randomised study of efmarodocokin alfa in healthy volunteers and patients with ulcerative colitis. Gut. 2023 Aug;72(8):1451-1461. doi: 10.1136/gutjnl-2022-328387. Epub 2023 Feb 2. PMID: 36732049.
- Danese S, Rothenberg ME, Lim JJ, Ding HT, McBride JM, Chen Y, et al. A randomized phase II study of efmarodocokin alfa, an interleukin-22 agonist, versus vedolizumab in patients with ulcerative colitis. Clin Gastroenterol Hepatol. 2025 Jul;23(8):1387-1397. doi: 10.1016/j.cgh.2024.11.013. Epub 2024 Dec 16. PMID: 39694207.
- Guttman-Yassky E, Brunner PM, Neumann AU, Khattri S, Pavel AB, Malik K, et al. Efficacy and safety of fezakinumab (an IL-22 monoclonal antibody) in adults with moderate-to-severe atopic dermatitis inadequately controlled by conventional treatments: a randomized, double-blind, phase 2a trial. J Am Acad Dermatol. 2018 May;78(5):872-881.e6. doi: 10.1016/j.jaad.2018.01.016. Epub 2018 Jan 17. PMID: 29353025.
- Ma J, Li X, Wang C. The application of nanomaterials in the treatment of pancreatic-related diseases. Int J Mol Sci. 2024 Dec 7;25(23):13158. doi: 10.3390/ijms252313158. PMID: 39684868.