Международный эндокринологический журнал Том 22, №3, 2026
Вернуться к номеру
Епігенетичний вплив довгих некодуючих РНК на розвиток метаболічно асоційованого стеатогепатиту
Авторы: A.Ye. Abaturov, A.O. Nikulina
Dnipro State Medical University, Dnipro, Ukraine
Рубрики: Эндокринология
Разделы: Справочник специалиста
Версия для печати
Актуальність. Довгі некодуючі РНК (long non-coding RNA — lncR) характеризуються високим ступенем тканиноспецифічної експресії генів, що дозволяє розглядати певні їх сукупності як діагностичні маркери метаболічно асоційованого стеатогепатиту (metabolic-associated steatohepatitis — MASH). Метою цього огляду є надати сучасні дані щодо ролі довгих некодуючих РНК в епігенетичному впливі на розвиток метаболічно асоційованого стеатогепатиту шляхом аналізу даних сучасної наукової літератури. Матеріали та методи. Проведено аналіз 76 літературних джерел баз даних MEDLINE, Embase, PreMedline In-Process & Other Non-Indexed Citations, The Cochrane Systematic Reviews Database, DARE, NHS EED та HTA, Web of Knowledge Science Citation Index, Web of Knowledge ISI Proceedings, CRD, BIOSIS за останні п’ять років, які були відібрані за ключовими словами: довгі некодуючі стеатогенні РНК; епігенетична регуляція; метаболічно асоційована жирова хвороба печінки; стеатотична хвороба печінки, асоційована з метаболічною дисфункцією; ожиріння. PROSPERO CRD420250652980. Аналіз літератури. Темпоральний аналіз показав, що періодичні флуктуації експресії більшості lncR призводять до розвитку як запалення, так і продукції прозапальних цитокінів. Інгібування фактора транскрипції NF-κB пригнічує активність експресії більшості прозапальних lncR. Довгі некодуючі РНК беруть участь у розвитку MASH переважно шляхом активації прозапальних сигнальних шляхів, факторів транскрипції (NF-κB, AP-1) та інфламасом. Довгі некодуючі РНК, регулюючи рівень експресії цитокінів (IL-1β, IL-6, TNF) та хемокінів (CCL2, CXCL1, CXCL5), визначають рекрутинг прозапальних імуноцитів, місцеву судинну реакцію і, як наслідок, ступінь запальної реакції тканини печінки при стеатотичній хворобі печінки, асоційованій із метаболічною дисфункцією. Висновки. Прозапальні й протизапальні lncR є епігенетичними регуляторами запалення печінки, що визначають розвиток MASH і можуть розглядатися як потенційні мішені протизапальної медикаментозної терапії хворих на MASH.
Background. Long non-coding RNAs (lncR) are characterized by a high degree of tissue-specific gene expression, allowing certain combinations to be considered as diagnostic markers of metabolic-associated steatohepatitis (MASH). The purpose of this literature review is to present current data on the role of long non-coding RNAs in the epigenetic influence on the development of metabolic-associated steatohepatitis, based on analysis of contemporary scientific literature. Materials and methods. An analysis of 76 literature sources from the past five years was conducted using the databases MEDLINE, Embase, PreMedline In-Process & Other Non-Indexed Citations, The Cochrane Systematic Reviews Database, DARE, NHS EED and HTA databases, Web of Knowledge Science Citation Index, Web of Knowledge ISI Proceedings, CRD databases, and BIOSIS. Sources were selected using the keywords: long non-coding steatogenic RNAs, epigenetic regulation, metabolically associated fatty liver disease, metabolic dysfunction-associated steatotic liver disease, obesity. PROSPERO registration: CRD420250652980. Literature analysis. Temporal analysis showed that periodic fluctuations in the expression of most lncR lead to both inflammation development and production of pro-inflammatory cytokines. Inhibition of the transcription factor NF-κB suppresses the expression activity of most pro-inflammatory lncR. Long non-coding RNAs participate in the development of MASH primarily by activating pro-inflammatory signaling pathways, transcription factors (NF-κB, AP-1), and inflammasomes. By regulating the expression levels of cytokines (IL-1β, IL-6, TNF) and chemokines (CCL2, CXCL1, CXCL5), lncR determine the recruitment of pro-inflammatory immune cells, local vascular response, and, consequently, the degree of inflammatory reaction in liver tissue in metabolic dysfunction-associated steatotic liver disease. Conclusions. Pro-inflammatory and anti-inflammatory lncR are epigenetic regulators of liver inflammation that determine the development of MASH and may be considered potential targets for anti-inflammatory pharmacotherapy in patients with MASH.
діти; ожиріння; метаболічно асоційований стеатогепатит; довгі некодуючі РНК; огляд літератури
children; obesity; metabolic-associated steatohepatitis; long non-coding RNAs; literature review
Для ознакомления с полным содержанием статьи необходимо оформить подписку на журнал.
- Ibrahim SH, Hirsova P, Gores GJ. Non-alcoholic steatohepa–titis pathogenesis: sublethal hepatocyte injury as a driver of liver inflammation. Gut. 2018 May;67(5):963-972. doi: 10.1136/gutjnl-2017-315691.
- Katsarou A, Moustakas II, Pyrina I, Lembessis P, Koutsilie–ris M, Chatzigeorgiou A. Metabolic inflammation as an instigator of fibrosis during non-alcoholic fatty liver disease. World J Gastroenterol. 2020 May 7;26(17):1993-2011. doi: 10.3748/wjg.v26.i17.1993.
- Hammerich L, Tacke F. Hepatic inflammatory responses in liver fibrosis. Nat Rev Gastroenterol Hepatol. 2023 Oct;20(10):633-646. doi: 10.1038/s41575-023-00807-x.
- Rinella ME, Lazarus JV, Ratziu V, et al.; NAFLD Nomenclature consensus group. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology. 2023 Dec 1;78(6):1966-1986. doi: 10.1097/HEP.0000000000000520.
- Yahoo N, Dudek M, Knolle P, Heikenwälder M. Role of immune responses in the development of NAFLD-associated liver cancer and prospects for therapeutic modulation. J Hepatol. 2023 Aug;79(2):538-551. doi: 10.1016/j.jhep.2023.02.033.
- Grajower MM. Nonalcoholic Steatohepatitis. JAMA. 2020 Sep 1;324(9):899. doi: 10.1001/jama.2020.10434.
- Samji NS, Heda R, Kovalic AJ, Satapathy SK. Similarities and Differences Between Nonalcoholic Steatohepatitis and Other Causes of Cirrhosis. Gastroenterol Clin North Am. 2020 Mar;49(1):151-164. doi: 10.1016/j.gtc.2019.09.004.
- Sheka AC, Adeyi O, Thompson J, Hameed B, Crawford PA, Ikramuddin S. Nonalcoholic Steatohepatitis: A Review. JAMA. 2020 Mar 24;323(12):1175-1183. doi: 10.1001/jama.2020.2298.
- Shah PA, Patil R, Harrison SA. NAFLD-related hepatocellular carcinoma: The growing challenge. Hepatology. 2023 Jan 1;77(1):323-338. doi: 10.1002/hep.32542.
- Chen J, Ao L, Yang J. Long non-coding RNAs in disea–ses related to inflammation and immunity. Ann Transl Med. 2019 Sep;7(18):494. doi: 10.21037/atm.2019.08.37.
- Walther K, Schulte LN. The role of lncRNAs in innate immunity and inflammation. RNA Biol. 2021 May;18(5):587-603. doi: 10.1080/15476286.2020.1845505.
- Wang W, Yang N, Yang YH, Wen R, Liu CF, Zhang TN. Non-Coding RNAs: Master Regulators of Inflammasomes in Inflammatory Diseases. J Inflamm Res. 2021 Sep 30;14:5023-5050. doi: 10.2147/JIR.S332840.
- Rashidmayvan M, Sahebi R, Ghayour-Mobarhan M. Long non-coding RNAs: a valuable biomarker for metabolic syndrome. Mol Genet Genomics. 2022 Sep;297(5):1169-1183. doi: 10.1007/s00438-022-01922-1.
- Zhang Y, Liu H, Niu M, Wang Y, Xu R, Guo Y, Zhang C. Roles of long noncoding RNAs in human inflammatory diseases. Cell Death Discov. 2024 May 15;10(1):235. doi: 10.1038/s41420-024-02002-6.
- Uchida S, Kauppinen S. Long Non-Coding RNAs in Liver Cancer and Nonalcoholic Steatohepatitis. Noncoding RNA. 2020 Aug 29;6(3):34. doi: 10.3390/ncrna6030034.
- Ashrafizadeh M, Zarrabi A, Mostafavi E, Aref AR, Sethi G, Wang L, Tergaonkar V. Non-coding RNA-based regulation of inflammation. Semin Immunol. 2022 Jan;59:101606. doi: 10.1016/j.smim.2022.101606.
- Abaturov OE, Nikulina AO. Characterization of anti-steatogenic long noncoding RNAs and their epigenetic influence on the development of metabolic fatty liver disease — a systematic review. Eur J Clin Exp Med. 2025;23(3):767-775. doi: 10.15584/ejcem.2025.3.16.
- Lu J, Guo J, Liu J, Mao X, Xu K. Long Non-coding RNA MALAT1: A Key Player in Liver Diseases. Front Med (Lausanne). 2022 Jan 25;8:734643. doi: 10.3389/fmed.2021.734643.
- Zeng Q, Liu CH, Wu D, Jiang W, Zhang N, Tang H. LncRNA and circRNA in Patients with Non-Alcoholic Fatty Liver Disease: A Systematic Review. Biomolecules. 2023 Mar 20;13(3):560. doi: 10.3390/biom13030560.
- Xia J, Xin L, Zhu W, Li L, Li C, Wang Y, et al. Characterization of long non-coding RNA transcriptome in high-energy diet induced nonalcoholic steatohepatitis minipigs. Sci Rep. 2016 Jul 28;6:30709. doi: 10.1038/srep30709.
- Ye L, Zhao D, Xu Y, Lin J, Xu J, Wang K, et al. LncRNA-Gm9795 promotes inflammation in non-alcoholic steatohepatitis via NF-κB/JNK pathway by endoplasmic reticulum stress. J Transl Med. 2021 Mar 9;19(1):101. doi: 10.1186/s12967-021-02769-7.
- Guo B, Yan S, Zhai L, Cheng Y. LncRNA HOTAIR accele–rates free fatty acid-induced inflammatory response in HepG2 cells by recruiting SRSF1 to stabilize MLXIPL mRNA. Cytotechnology. 2024 Apr;76(2):259-269. doi: 10.1007/s10616-023-00614-x.
- Chen Y, Wang YJ, Zhao Y, Wang JC. Carbohydrate response element binding protein (ChREBP) modulates the inflammatory response of mesangial cells in response to glucose. Biosci Rep. 2018 Dec 7;38(6):BSR20180767. doi: 10.1042/BSR20180767.
- Chini A, Guha P, Malladi VS, Guo Z, Mandal SS. Novel long non-coding RNAs associated with inflammation and macrophage activation in human. Sci Rep. 2023 Mar 10;13(1):4036. doi: 10.1038/s41598-023-30568-1.
- Shen X, Guo H, Xu J, Wang J. Inhibition of lncRNA HULC improves hepatic fibrosis and hepatocyte apoptosis by inhibiting the MAPK signaling pathway in rats with nonalcoholic fatty liver disease. J Cell Physiol. 2019 Aug;234(10):18169-18179. doi: 10.1002/jcp.28450.
- Yu X, Zheng H, Chan MT, Wu WK. HULC: an oncoge–nic long non-coding RNA in human cancer. J Cell Mol Med. 2017 Feb;21(2):410-417. doi: 10.1111/jcmm.12956.
- Sallam T, Jones MC, Gilliland T, Zhang L, Wu X, Eskin A, et al. Feedback modulation of cholesterol metabolism by the lipid-responsive non-coding RNA LeXis. Nature. 2016 Jun 2;534(7605):124-8. doi: 10.1038/nature17674.
- Rossi A, Moro A, Tebaldi T, Cornella N, Gasperini L, Lunelli L, et al. Identification and dynamic changes of RNAs isolated from RALY-containing ribonucleoprotein complexes. Nucleic Acids Res. 2017 Jun 20;45(11):6775-6792. doi: 10.1093/nar/gkx235.
- Park JG, Kim G, Jang SY, Lee YR, Lee E, Lee HW, et al. Plasma Long Noncoding RNA LeXis is a Potential Diagnostic Marker for Non-Alcoholic Steatohepatitis. Life (Basel). 2020 Oct 3;10(10):E230. doi: 10.3390/life10100230.
- Du G, Jiang Z, Xia T, Liu M, Liu Z, Zhou H, et al. lincRNA00907 promotes NASH progression by targeting miRNA-942-5p/TAOK1. Aging (Albany NY). 2024 Apr 10;16(8):6868-6882. doi: 10.18632/aging.205730.
- Zhu L, Yu Q, Gao P, Liu Q, Luo X, Jiang G, et al. TAOK1 positively regulates TLR4-induced inflammatory responses by promoting ERK1/2 activation in macrophages. Mol Immunol. 2020 Apr 25;122:124-131. doi: 10.1016/j.molimm.2020.04.006.
- Zhang Z, Tang Z, Ma X, Sun K, Fan L, Fang J, et al. TAOK1 negatively regulates IL-17-mediated signaling and inflammation. Cell Mol Immunol. 2018 Aug;15(8):794-802. doi: 10.1038/cmi.2017.158.
- Yu L, Yang C, Shang N, Ding H, Zhu J, Zhu Y, et al. Paternal De Novo Variant of TAOK1 in a Fetus With Structural Brain Abnormalities. Front Genet. 2022 Jul 19;13:836853. doi: 10.3389/fgene.2022.836853.
- Atanasovska B, Rensen SS, Marsman G, Shiri-Sverdlov R, Withoff S, Kuipers F, et al. Long Non-Coding RNAs Involved in Progression of Non-Alcoholic Fatty Liver Disease to Steatohepatitis. Cells. 2021 Jul 25;10(8):1883. doi: 10.3390/cells10081883.
- Lei L, Chen J, Huang J, Lu J, Pei S, Ding S, et al. Functions and regulatory mechanisms of metastasis-associated lung adenocarcinoma transcript 1. J Cell Physiol. 2018 Jan;234(1):134-151. doi: 10.1002/jcp.26759.
- Biswas S, Thomas AA, Chen S, Aref-Eshghi E, Feng B, Gonder J, et al. MALAT1: An Epigenetic Regulator of Inflammation in Diabetic Retinopathy. Sci Rep. 2018 Apr 25;8(1):6526. doi: 10.1038/s41598-018-24907-w.
- Puthanveetil P, Chen S, Feng B, Gautam A, Chakrabarti S. Long non-coding RNA MALAT1 regulates hyperglycaemia induced inflammatory process in the endothelial cells. J Cell Mol Med. 2015 Jun;19(6):1418-25. doi: 10.1111/jcmm.12576.
- Leti F, Legendre C, Still CD, Chu X, Petrick A, Gerhard GS, DiStefano JK. Altered expression of MALAT1 lncRNA in nonalcoholic steatohepatitis fibrosis regulates CXCL5 in hepatic stellate cells. Transl Res. 2017 Dec;190:25-39.e21. doi: 10.1016/j.trsl.2017.09.001.
- Hwang S, Yun H, Moon S, Cho YE, Gao B. Role of Neutrophils in the Pathogenesis of Nonalcoholic Steatohepatitis. Front Endocrinol (Lausanne). 2021 Oct 11;12:751802. doi: 10.3389/fendo.2021.751802.
- Lauszus JS, Eriksen PL, Hansen MM, Eriksen LL, Shawcross DL, Vilstrup H, et al. Activation and Functional Priming of Blood Neutrophils in Non-Alcoholic Fatty Liver Disease Increases in Non-Alcoholic Steatohepatitis. Clin Exp Gastroenterol. 2021 Nov 13;14:441-449. doi: 10.2147/CEG.S329424.
- Scott C, Stokes R, Cha KM, Clouston A, Eslam M, Metwally M, et al. Myeloid cell deletion of Aryl hydrocarbon Receptor Nuclear Translocator (ARNT) induces non-alcoholic steatohepatitis. PLoS One. 2019 Dec 4;14(12):e0225332. doi: 10.1371/journal.pone.0225332.
- Xiang J, Deng YY, Liu HX, Pu Y. LncRNA MALAT1 Promotes PPARα/CD36-Mediated Hepatic Lipogenesis in Nonalcoholic Fatty Liver Disease by Modulating miR-206/ARNT Axis. Front Bioeng Biotechnol. 2022 Jun 13;10:858558. doi: 10.3389/fbioe.2022.858558.
- Qiu YY, Zhang J, Zeng FY, Zhu YZ. Roles of the peroxisome proliferator-activated receptors (PPARs) in the pathogenesis of nonalcoholic fatty liver disease (NAFLD). Pharmacol Res. 2023 Jun;192:106786. doi: 10.1016/j.phrs.2023.106786.
- Chinetti G, Fruchart JC, Staels B. Peroxisome proliferator-activated receptors and inflammation: from basic science to clinical applications. Int J Obes Relat Metab Disord. 2003 Dec;27 Suppl 3:S41-5. doi: 10.1038/sj.ijo.0802499.
- Bougarne N, Weyers B, Desmet SJ, Deckers J, Ray DW, Staels B, De Bosscher K. Molecular Actions of PPARα in Lipid Metabolism and Inflammation. Endocr Rev. 2018 Oct 1;39(5):760-802. doi: 10.1210/er.2018-00064.
- Carambia A, Schuran FA. The aryl hydrocarbon receptor in liver inflammation. Semin Immunopathol. 2021 Aug;43(4):563-575. doi: 10.1007/s00281-021-00867-8.
- Long F, Zhou X, Zhang J, Di C, Li X, Ye H, et al. The role of lncRNA HCG18 in human diseases. Cell Biochem Funct. 2024 Mar;42(2):e3961. doi: 10.1002/cbf.3961.
- Chen L, Li C, Peng Z, Zhao J, Gong G, Tan D. miR-197 Expression in Peripheral Blood Mononuclear Cells from Hepatitis B Virus-Infected Patients. Gut Liver. 2013 May;7(3):335-42. doi: 10.5009/gnl.2013.7.3.335.
- Zhang L, Zhou X, Dai Y, Lv C, Wu J, Wu Q, et al. Establishment of interleukin-18 time-resolved fluorescence immunoassay and its preliminary application in liver disease. J Clin Lab Anal. 2021 May;35(5):e23758. doi: 10.1002/jcla.23758.
- Xia Y, Zhang Y, Wang H. Upregulated lncRNA HCG18 in Patients with Non-Alcoholic Fatty Liver Disease and Its Regulatory Effect on Insulin Resistance. Diabetes Metab Syndr Obes. 2021 Dec 3;14:4747-4756. doi: 10.2147/DMSO.S333431.
- Pan Y, Wang T, Zhao Z, Wei W, Yang X, Wang X, Xin W. Novel Insights into the Emerging Role of Neat1 and Its Effects Downstream in the Regulation of Inflammation. J Inflamm Res. 2022 Jan 26;15:557-571. doi: 10.2147/JIR.S338162.
- Chen S, Wang J, Zhang K, Ma B, Li X, Wei R, Nian H. LncRNA Neat1 targets NonO and miR-128-3p to promote antigen-speci–fic Th17 cell responses and autoimmune inflammation. Cell Death Dis. 2023 Sep 16;14(9):610. doi: 10.1038/s41419-023-06132-0.
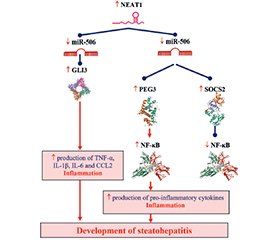
- Jin SS, Lin XF, Zheng JZ, Wang Q, Guan HQ. lncRNA NEAT1 regulates fibrosis and inflammatory response induced by nonalcoholic fatty liver by regulating miR-506/GLI3. Eur Cytokine Netw. 2019 Sep 1;30(3):98-106. doi: 10.1684/ecn.2019.0432.
- Matissek SJ, Karbalivand M, Han W, Boutilier A, Yzar-Garcia E, Kehoe LL, et al. A novel mechanism of regulation of the oncoge–nic transcription factor GLI3 by toll-like receptor signaling. Oncotarget. 2022 Aug 3;13:944-959. doi: 10.18632/oncotarget.28261.
- Zhang Z, Wen H, Peng B, Weng J, Zeng F. Downregulated microRNA-129-5p by Long Non-coding RNA NEAT1 Upregulates PEG3 Expression to Aggravate Non-alcoholic Steatohepatitis. Front Genet. 2021 Jan 26;11:563265. doi: 10.3389/fgene.2020.563265.
- Ye J, Lin Y, Yu Y, Sun D. LncRNA NEAT1/micro–RNA-129-5p/SOCS2 axis regulates liver fibrosis in alcoholic steatohepatitis. J Transl Med. 2020 Nov 24;18(1):445. doi: 10.1186/s12967-020-02577-5.
- Abaturov OE, Nikulina AO. Epigenetic influence of long non-coding RNAs on the development of insulin resistance in metaboli–cally associated fatty liver disease (part 2). International Journal of Endocrinology (Ukraine). 2025;21(3):322-331. doi: 10.22141/2224-0721.21.3.2025.1545.
- Li S, Han S, Jin K, Yu T, Chen H, Zhou X, et al. SOCS2 Suppresses Inflammation and Apoptosis during NASH Progression through Limiting NF-κB Activation in Macrophages. Int J Biol Sci. 2021 Oct 11;17(15):4165-4175. doi: 10.7150/ijbs.63889.
- Zhang P, Cao L, Zhou R, Yang X, Wu M. The lncRNA Neat1 promotes activation of inflammasomes in macrophages. Nat Commun. 2019 Apr 2;10(1):1495. doi: 10.1038/s41467-019-09482-6.
- Bertran L, Eigbefoh-Addeh A, Portillo-Carrasquer M, Barrientos-Riosalido A, Binetti J, Aguilar C, et al. Identification of the Potential Molecular Mechanisms Linking RUNX1 Activity with Nonalcoholic Fatty Liver Disease, by Means of Systems Biology. Biomedi–cines. 2022 Jun 3;10(6):1315. doi: 10.3390/biomedicines10061315.
- Kaur S, Rawal P, Siddiqui H, Rohilla S, Sharma S, Tripathi DM, et al. Increased Expression of RUNX1 in Liver Correlates with NASH Activity Score in Patients with Non-Alcoholic Steatohepatitis (NASH). Cells. 2019 Oct 18;8(10):1277. doi: 10.3390/cells8101277.
- Ma TT, Huang C, Ni Y, Yang Y, Li J. ATP Citrate Lyase and LncRNA NONMMUT010685 Play Crucial Role in Nonalcoholic Fatty Liver Disease Based on Analysis of Microarray Data. Cell Physiol Biochem. 2018;51(2):871-885. doi: 10.1159/000495384.
- Wang Q, Zhou H, Bu Q, Wei S, Li L, Zhou J, et al. Role of XBP1 in regulating the progression of non-alcoholic steatohepatitis. J Hepatol. 2022 Aug;77(2):312-325. doi: 10.1016/j.jhep.2022.02.031.
- Wang Q, Bu Q, Liu M, Zhang R, Gu J, Li L, et al. XBP1-mediated activation of the STING signalling pathway in macrophages contributes to liver fibrosis progression. JHEP Rep. 2022 Aug 18;4(11):100555. doi: 10.1016/j.jhepr.2022.100555.
- Feltham R, Silke J. The small molecule that packs a punch: ubiquitin-mediated regulation of RIPK1/FADD/caspase-8 complexes. Cell Death Differ. 2017 Jul;24(7):1196-1204. doi: 10.1038/cdd.2017.67.
- Malireddi RKS, Kesavardhana S, Kanneganti TD. ZBP1 and TAK1: Master Regulators of NLRP3 Inflammasome/Pyroptosis, Apoptosis, and Necroptosis (PAN-optosis). Front Cell Infect Microbiol. 2019 Nov 26;9:406. doi: 10.3389/fcimb.2019.00406.
- Li W, Yuan J. Targeting RIPK1 kinase for modulating inflammation in human diseases. Front Immunol. 2023 Mar 8;14:1159743. doi: 10.3389/fimmu.2023.1159743.
- Tao L, Yi Y, Chen Y, Zhang H, Orning P, Lien E, et al. RIP1 kinase activity promotes steatohepatitis through mediating cell death and inflammation in macrophages. Cell Death Differ. 2021 Apr;28(4):1418-1433. doi: 10.1038/s41418-020-00668-w.
- Majdi A, Aoudjehane L, Ratziu V, Islam T, Afonso MB, Conti F, et al. Inhibition of receptor-interacting protein kinase 1 improves experimental non-alcoholic fatty liver disease. J Hepatol. 2020 Apr;72(4):627-635. doi: 10.1016/j.jhep.2019.11.008.
- Shabgah AG, Norouzi F, Hedayati-Moghadam M, Soleimani D, Pahlavani N, Navashenaq JG. A comprehensive review of long non-coding RNAs in the pathogenesis and development of non-alcoholic fatty liver disease. Nutr Metab (Lond). 2021 Feb 23;18(1):22. doi: 10.1186/s12986-021-00552-5.
- Atanasovska B, Rensen SS, van der Sijde MR, Marsman G, Kumar V, Jonkers I, et al. A liver-specific long noncoding RNA with a role in cell viability is elevated in human nonalcoholic steatohepatitis. Hepatology. 2017 Sep;66(3):794-808. doi: 10.1002/hep.29034.
- Wei L, He P, Tan Z, Zhao L, Lin C, Wei Z. Unveiling the role of the KLF4/Lnc18q22.2/ULBP3 axis in the tumorigenesis and immune escape of hepatocellular carcinoma under hypoxic condition. J Cell Mol Med. 2024 May;28(10):e18411. doi: 10.1111/jcmm.18411.
- Lin Y, Wang S, Gao L, Zhou Z, Yang Z, Lin J, et al. Oscilla–ting lncRNA Platr4 regulates NLRP3 inflammasome to ameliorate nonalcoholic steatohepatitis in mice. Theranostics. 2021 Jan 1;11(1):426-444. doi: 10.7150/thno.50281.
- Chen Y, Chen X, Gao J, Xu C, Xu P, Li Y, et al. Long noncoding RNA FLRL2 alleviated nonalcoholic fatty liver disease through Arntl-Sirt1 pathway. FASEB J. 2019 Oct;33(10):11411-11419. doi: 10.1096/fj.201900643RRR.
- Meng D, Zhang F, Yu W, Zhang X, Yin G, Liang P, et al. Bio–logical Role and Related Natural Products of SIRT1 in Nonalcoholic Fatty Liver. Diabetes Metab Syndr Obes. 2023 Dec 8;16:4043-4064. doi: 10.2147/DMSO.S437865.
- Gadallah SH, Eissa S, Ghanem HM, Ahmed EK, Hasa–nin AH, El Mahdy MM, Matboli M. Probiotic-prebiotic-synbiotic modulation of (YAP1, LATS1 and NF2 mRNAs/miR-1205/lncRNA SRD5A3-AS1) panel in NASH animal model. Biomed Pharmacother. 2021 Aug;140:111781. doi: 10.1016/j.biopha.2021.111781.