Международный неврологический журнал Том 18, №1, 2022
Діагностика і лікування астроцитоми: огляд літератури
Авторы: Dipak Chaulagain, Volodymyr Smolanka, Andriy Smolanka
Regional Clinical Center of Neurosurgery and Neurology, Uzhhorod National University, Uzhhorod, Ukraine
Рубрики: Неврология
Разделы: Клинические исследования
Резюме
Астроцитома є досить поширеною формою пухлини головного мозку. Вона розвивається з астроцитів — зіркоподібних клітин, що утворюють «клейоподібну» або підтримуючу тканину. Астроцитоми класифікують за характером і тяжкістю. За даними Всесвітньої організації охорони здоров’я, виділяють I–IV ступені цих пухлин. Пілоцитарна астроцитома, астроцитома II ступеня, анапластична астроцитома і мультиформна гліобластома є найпоширенішими видами астроцитоми. Однак залежно від виду клінічні прояви і діагноз можуть змінюватися. Хірургічне лікування теж відрізняється залежно від типу астроцитоми. Мета цього дослідження: визначити основні характеристики різних форм астроцитоми, краще зрозуміти діагностику і терапію, а також провести пошук публікацій у базах даних NCBI, PubMed та Google Scholar. У статті наведено огляд лікування астроцитоми на основі її типу; результати значною мірою залежать від різних форм астроцитоми, можливості виконати тотальну резекцію і, нарешті, прогнозу. Знання типів астроцитоми і проявів може допомогти при хірургічному лікуванні. Ми також представили огляд літератури щодо типів і діагностики астроцитоми та оцінили іcнуючі хірургічні методи лікування та прогноз.
Astrocytoma is a form of a brain tumor that is rather prevalent. It originates in astrocytes, star-shaped cells that make up the “glue-like” or supporting tissue. Astrocytomas are classified by their nature and severity. According to the World Health Organization, there are I–IV grades of these tumours. Pilocytic astrocytoma, grade II astrocytoma, anaplastic astrocytoma, and glioblastoma multiforme are the most common types of astrocytoma. However, depending on the type, its clinical appearance and diagnosis may change. Similarly, surgical management differs depending on the type of astrocytoma. The current study aims to identify the basic characteristics of different forms of astrocytoma, better understand diagnosis and therapy, and search for publications in NCBI, PubMed, and Google Scholar databases. This article provides an overview of astrocytoma therapy based on its type; the findings greatly depend on different forms of astrocytoma, possibility to perform gross total resection, and lastly, the prognosis. Knowledge of astrocytoma types and manifestations may help in the surgical management of astrocytoma. We also presented a review of the literature on the types and diagnosis of astrocytoma and evaluated current surgical treatments and prognosis.
Ключевые слова
астроцитома; огляд; діагностика; обсяг резекції; хірургічне лікування
astrocytoma; review; diagnosis; extent of resection; surgical management
Introduction
According to Brain Tumour Charity (2018), astrocytoma is a tumour that develops from a type of brain cell called an astrocyte. Throughout the spinal cord and brain, there are nerve cells known as neurons that transmit messages to, within and from the brain. Surrounding the neurons are cells known as glial cells that support and protect the neurons by providing them with nutrients and oxygen and removing dead cells. An astrocyte secures and assists neurons which also help to pass information between neurons and it is also essential in processing the messages in brain.
Kabel et al. (2018) has stated that any tumour which emerges from brain’s supportive or glial tissue is known as glioma. One type of glioma is known as astrocytoma. Astrocytomas are named after astrocytes, the star-shaped cells from which they grow. The accurate cause of astrocytoma is not known and it occurs more often in men than in women. Astrocytomas can develop both in adults and children. The treatment methods used for astrocytomas are chemotherapy, surgery, radiation therapy or combination of treatments.
Moini and Piran (2020) have stated that the tumours which emerge from astrocytes are known as astrocytomas. The tumour may be malignant or benign and it is graded based on abnormal or normal cell qualities. The astrocytomas may exist anywhere in the central nervous system. The early symptoms of astrocytomas are seizures, headaches, memory loss and changes in behaviour. The treatments used for astrocytomas include radiation therapy, surgery, follow-up scan, survey and chemotherapy.
According to Kapoor and Gupta (2021), astrocytoma originates in astrocytes which are a type of glial star-shaped cells in the cerebrum. Astrocytoma is the most common glioma which generally affects the brain of a human being and sometimes also develops in the spinal cord. Glial tumours are the common cause of morbidity and mortality in both old and young age people. One of the common brain tumours is astrocytoma.
Moini et al. (2021) has mentioned that the common form of glioma is astrocytoma. The most common symptoms of astrocytomas are headache due to increased intracranial pressure, loss of memory, seizures, changes in behaviour, head tilt, neck stiffness, incoordination, vomiting, nausea, visual problems and irritability. The treatment method for astrocytomas is a combination of radiation therapy, surgery, follow-up imaging, observation, ventricular shunting and chemotherapy.
Astrocytoma types
According to the research of Hirtz et al. (2020), there are four types of astrocytoma, from grade I to grade IV. Grade I tumour is known as pilocytic astrocytoma which is always benign and can be resected. Grade II tumours are diffuse astrocytomas, low-grade or infiltrative gliomas. They deve-lop gradually that will acquire a much aggressive phenotype over time. Grade III tumours are anaplastic astrocytomas including oligoastrocytomas and oligodendrogliomas. Grade IV tumour, glioblastoma multiforme, is characterized by a rapidly developing phenotype with the presence of necrotic areas and vascular growth.
Bikowska-Opalach et al. (2014) have described diffuse astrocytomas as grade II astrocytomas which occur in young people though they also affect senior patients and children. They may develop in any part of the brain but are most typical for hemispheres of the cerebrum. The diffuse astrocytomas tend to develop into surrounding normal tissue of brain. Headaches, weakness in one part of the body and seizures are the earliest symptoms of brain tumour.
Grimm and Chamberlain (2016) have reported that anaplastic astrocytoma is a malignant, diffusely infiltrating primary brain tumour with a median onset of 41 years of age. Anaplastic astrocytoma is defined by histological features of increased cellularity, nuclear atypia, significant activity of proliferation as manifested in mitoses and lacking either necrosis or endothelial proliferation which are the two hallmarks of glioblastoma.
Singla and Gupta (2016) have described grade IV astrocytoma which is named glioblastoma multiforme. It is a malignant tumour which increasingly spreads to other brain parts. There are two types of grade IV astrocytoma, primary and secondary. Primary astrocytoma is the most common and most aggressive whereas secondary astrocytoma is a low-grade tumour that develops into grade IV tumours.
In the research of Volovetz et al. (2019), it is stated that pilocytic astrocytoma is a type of astrocytoma with specific genetic, histological and clinical features. It is the most frequent primary brain tumour which affects mainly adolescents and children though it may also present in adulthood.
Table 1 shows the reviews of the types of astrocytoma.
Diagnosis of аstrocytoma
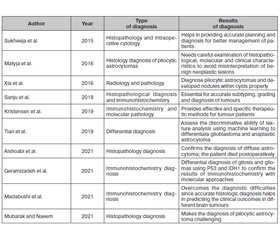
Sukheeja et al. (2015) have stated in their research that the prognosis assessment by grading astrocytoma has been challenging activity for pathologists though surgical and radiological imaging approaches have improved significantly. This research has attempted to assess tumours intraoperatively for quick diagnosis. MIB-1 labelling index was used to study their proliferative activity. Intraoperative cytology is useful and accurate in consultation of intraoperation. MIB-1 labelling index was used as a conjunct in this research for grading in little biopsies.
In the work of Matyja et al. (2016), the prognosis and behaviour of pilocytic astrocytomas correlated with location and histology of tumours. Pilomyxoid astrocytoma is a classical pilocytic astrocytoma with greater invasive development and increased risk of dissemination and recurrences. Pilocytic astrocytomas have varied histology with biphasic architecture of loose, compact and microcystic fibrillary regions. The pilocytic astrocytoma diagnosis requires careful examination of histopathological, molecular and clinical characteristics to avoid misinterpretation of benign neoplastic lesions.
The main purpose of Xia et al. (2016) study was to recognize imaging features of pilocytic astrocytomas in ventricles of the cerebrum to help radiologists differentiate pilocytic astrocytomas and other brain tumours preoperatively. The radiological and clinical records were analysed. The radiological characteristics of extraventricular and intraventricular pilocytic astrocytomas were similar to typical ones including developed nodules within cysts.
Sanju et al. (2018) have mentioned in their research that brain tumours are heterogeneous neoplasms with annual incidence of 5 to 10 % per 100,000 population in India. The tumours classification is an art of pathology to differentiate malignant and benign lesions on histological basis. This research reveals that histopathology is an essential component for grading and diagnosis in some cases. Immunohistoche-mistry plays an essential role in critical cases where grading and diagnosis are not feasible only on the basis of histology.
Kristensen et al. (2019) propose an integrated histomolecular method which allows for an accurate diagnosis of embryonal central nervous system tumours and diffuse gliomas. This study provides an update of several essential prognostic markers and diagnostic markers for state-of-the-art identification of primary central nervous system tumours. Immunohistochemistry is a useful tool for characterizing molecules of many brain tumours.
Tian et al. (2019) have described the two most common primary brain tumours in adults, anaplastic astrocytoma and glioblastoma. The differential diagnosis is essential for prognosis and treatment suggestions. The main aim of the study is to assess the discriminative ability of texture analysis using machine learning to differentiate glioblastoma and anaplastic astrocytoma. The texture characteristics were taken from contrast-enhanced magnetic resonance imaging using the LifeX package. The results of the research show that magnetic resonance imaging using linear discriminant analysis algorithm had promising ability to distinguish glioblastoma and anaplastic astrocytoma.
Alshoabi et al. (2021) provided a case report of diffuse astrocytoma and the diagnostic dilemma in an unusual phenotype. Diffuse astrocytoma is a glioma of infiltrating type which is difficult to diagnose even with histopathology. The results of histopathology confirmed the identification of diffuse astrocytoma; the patient died postoperatively.
Geramizadeh et al. (2021) presented a study on a simple panel of isocitrate dehydrogenase 1 (IDH1) and P53 in differential diagnosis of low-grade astrocytoma and reactive gliosis. Reactive gliosis is a glial tissue response to various types of injury namely trauma, abscess, neoplastic or haemorrhage process of the brain. In certain situations, particularly when the biopsy of tissue is small, there may be difficulty to discriminate the reactive condition with low-grade diffuse astrocytoma by hematoxylin and eosin slides, so some molecular and immunohistochemical markers have been introduced for differential diagnosis. In this research, the authors evaluated P53 and IDH1 mutation by immunohistochemistry as a highly sensitive, easy and specific approach to distinguish reactive gliosis and low-grade astrocytoma. The combination of IDH1 and P53 as an immunohistochemical panel reveals 91% sensitivity and 96% specificity for differential diagnosis of low-grade astrocytoma and reactive gliosis. These two markers can be useful for differential diagnosis.
In the research of Madabushi et al. (2021), central nervous system tumours comprise 1 to 2 % of all tumours which pose diagnostic difficulties because various histogenesis of tumours reveals different overlap and differentiation in morphological characteristics. The results of the research show that astrocytomas in adults were most common followed by nerve sheath tumours, meningiomas, metastatic deposits, gliosarcomas and glioblastomas. Tumour grading was performed as per revised criteria of the World Health Organization (WHO). Immunohistochemistry results were studied in selective cases. This research highlights the immunohistochemistry utility as an adjunct in histologic diagnosis of brain tumours in critical cases.
Mubarak and Naeem (2021) reviewed five cases in patients with histopathological diagnosis of pilocytic astrocytoma. Pilocytic astrocytoma is a low-grade glial tumour which occurs commonly in paediatric population. It is rare in adults and usually has better prognosis. These tumours have various locations in the central nervous system inclu-ding the fourth ventricle, cerebellum, optic chiasm, suprasellar area, brain stem and cerebral hemisphere in different patients. Pilocytic astrocytoma has a wide spectrum vast set of neuroradiological presentation. Besides its classical exi-stence as low-grade glioma, a more typical presentation makes the diagnosis challenging.
Table 2 shows the reviews of diagnosis of astrocytomas.
Treatment of astrocytoma
Barker et al. (2014) describe survival of patients treated with radiation therapy for anaplastic astrocytoma. Anaplastic astrocytomas make up 7 % of primary brain tumours in adults. This study assessed the association of patient-, treatment- and tumour-related factors with survival in astrocytoma. Medical records of patients with astrocytoma who underwent radiotherapy between 1987 and 2007 were reviewed. In this study, the classification of Radiation Therapy Oncology Group recursive partitioning analysis correlated with survival.
Forst et al. (2014) have mentioned in their study that low-grade gliomas are a diverse group of primary brain tumours that emerge in healthy patients and have an indolent course with higher survival compared to high-grade gliomas. The treatments include surgery, observation, chemotherapy, radiation or combination method and the management depends on tumour location, molecular profile, histology and patient characteristics. This study reviews current strategies for low-grade gliomas such as radiotherapy, chemotherapy and surgery.
Pace et al. (2014) have stated that patients with brain tumour differ from other cancer populations due to complexity of supportive care requirements, disease course, short life expectancy and resulting need for a particular palliative method. This research proposed a pilot model of palliative care for brain tumour patients.
According to Walbert and Chasteen (2015), brain tumour diagnosis is a life-changing situation for families and patients. This study discusses screening tools specific for symptoms and validates global symptom assessments to re-cognize distressing symptoms. Fatigue, seizure, anxiety and depression are common symptoms throughout the disease and must be managed properly.
Narita (2018) et al. proposed chemotherapy treatment for adults with diffuse astrocytoma. The chemoradiotherapy or chemotherapy efficiency in diffuse astrocytoma has been largely unknown until recently. Howe-ver, a randomized controlled study revealed that addition of CCNU, vincristine and procarbazine chemotherapy to fractioned radiotherapy in patients with high-risk WHO grade II gliomas, including those with diffuse astrocytoma, has essential positive effect on overall and progression-free survival. Temozolomide efficiency in low-grade gliomas was reported and a randomized phase III study that compares fractioned radiotherapy alone or in combination with temozolomide in unresectable diffuse astrocytoma is ongoing presently.
The research of Wang and Li (2018) presents synthesis of the most recent evidence of surgery and its efficacy in the treatment of astrocytoma patients. The findings of this study provide useful reference for the complications and efficiency of surgery in astrocytoma patients to future researchers and clinicians.
According to Wahner et al. (2020), current guidelines suggest maximal safe resection followed by chemotherapy and radiotherapy for anaplastic astrocytoma patients. They have a limited life expectancy despite this multimodal treatment method. In this study, variables related to the overall survival were identified and a score was built to find the overall survival of anaplastic astrocytoma patients at primary diagnosis stage. Implementing molecular mar-kers into the classification of the World Health Organization raised queries about the importance of clinical trials and grading revealed advantages of overall survival for combined radiochemotherapy.
Table 3 shows the reviews of the treatment types for astrocytoma.
Reviews on astrocytoma studies
Hervey-Jumper and Berger (2014) have stated that central nervous system tumour is a major cause of mortality and morbidity in the US. Outside of brain metastasis high- and low-grade gliomas are the most common intrinsic brain tumours. High-grade gliomas are aggressive with the vast majority of patients facing recurrence and a median survival of one to three years.
Xia et al. (2018) have mentioned in their research that resection is essential to conduct a pathological biopsy and to achieve intracranial pressure reduction in patients with low-grade gliomas. The main purpose of the study is to determine whether a greater extent of resection would improve the 5- and 10-year overall survival of these patients.
Patel et al. (2019) presented a study on the extent of surgical resection in lower-grade gliomas with differential impact based on molecular subtype. The diffuse low-grade gliomas are classified into molecular subtypes.
Wang et al. (2019) performed a systematic review on the advance of extent of resection in glioblastoma surgical treatment. This study summarizes and reviews the history of surgical resection for glioblastoma.
Choi et al. (2020) evaluated prognostic factors in adult low-grade gliomas according to the 2016 World Health Organization classification. Records of 153 patients diagnosed with WHO grade II low-grade glioma between 2003 and 2015 were reviewed retrospectively.
Laurent et al. (2020) carried out a retrospective analysis to assess the relationship between extent of resection and the occurrence of hospital-acquired conditions and patient safety indicators. The extent of resection is well established as correlating with overall survival in patients with glioblastoma.
The study of Mao et al. (2020) examines the relationship between survival results and surgical approaches in astrocytoma patients. People diagnosed with astrocytoma between 2004 and 2015 were identified using the Surveillance, Epidemiology and End Results database.
Karschnia et al. (2021) developed evidence-based expert recommendations on the types for extent of resection. Surgical resection is the standard of care in diffuse gliomas and more extensive tumour resection seems to be associated with favourable outcomes.
The main purpose of Motomura et al. (2021) study is to assess the impact of the extent of tumour resection on survival in patients with lower-grade gliomas who underwent awake brain mapping.
Revilla-Pacheco et al. (2021) proposed a systematic review and meta-analysis on extent of resection and survival in patients with glioblastoma multiforme. Glioblastoma multiforme has an ominous prognosis with overall survival of 14 months. The extent of surgical resection is one of the factors associated with better prognosis.
Table 4 shows the reviews of studies conducted on astrocytoma.
Conclusions
With the present growth in the treatment of brain astrocytoma, it is debated highly whether the extent of resection influences the survival. The maximum extent of resection must be tried safely while reducing the surgically-induced neurological risk. Genetics is the upcoming sector for brain tumour therapies. Various treatment methods are used to cure astrocytoma. Thus, it can be summarized that astrocytoma can be managed with greater quality clinical trials prospectively.
Received 04.01.2022
Revised 17.01.2022
Accepted 25.01.2022
Список литературы
1. Guram K., Smith M., Ginader T., Bodeker K., Pelland D., Pennington E., Buatti J.M. Using smaller-than-standard radiation treatment margins does not change survival outcomes in patients with high-grade gliomas. Pract. Radiat. Oncol. 2019 Jan. 9(1). 16-23. doi: 10.1016/j.prro.2018.06.001.
2. Kabel A.M., Modais K., Salim A., Ahmad R., Ahmad A., Alnumari K.A. Astrocytoma: insights into risk factors, pathogenesis, diagnosis and management. Journal of Cancer Research and Treatment. 2018. 6(3). 70-73. doi: 10.12691/jcrt-6-3-2.
3. Moini J., Piran P. Functional and Clinical Neuroanatomy. A Guide for Health Care Professionals. Ch. 1: Histophysiology. Elsevier, 2020. 1-49.
4. Kapoor M., Gupta V. Astrocytoma. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. 2021 Jan.
5. Moini J., Avgeropoulos N.G., Samsam M. Epidemiology of Brain and Spinal Tumours. Ch. 6: Astrocytoma. Elsevier Private Limited, 2021.
6. Hirtz A., Rech F., Dubois-Pot-Schneider H., Dumond H. Astrocytoma: a hormone-sensitive tumor? Int. J. Mol. Sci. 2020 Nov 30. 21(23). 9114. doi: 10.3390/ijms21239114.
7. Bikowska-Opalach B., Szlufik S., Grajkowska W., Jozwiak J. Pilocytic astrocytoma: a review of genetic and molecular factors, diagnostic and prognostic markers. Histol. Histopathology. 2014. 29(10). 1235-1248.
8. Grimm S.A., Chamberlain M.C. Anaplastic astrocytoma. CNS Oncol. 2016 Jul. 5(3). 145-57. doi: 10.2217/cns-2016-0002.
9. Singla C., Gupta S. Grade identification of astrocytoma using image processing: a literature review. Proceedings of 10th INDIACom. 2016. 8229.
10. Tork C.A., Atkinson C. Oligodendroglioma. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. 2021 Jan.
11. Sukheeja D., Singhvi S., Rai N.N., Midya M. A comparative study of histopathology of astrocytomas with intraoperative cytology with special reference to MIB-1 labelling index. J. Clin. Diagn. Res. 2015 Aug. 9(8). EC01-3. doi: 10.7860/JCDR/2015/12372.6281.
12. Matyja E., Grajkowska W., Stępień K., Naganska E. Heterogeneity of histopathological presentation of pilocytic astrocytoma — diagnostic pitfalls. A review. Folia Neuropathol. 2016. 54(3). 197-211. doi: 10.5114/fn.2016.62530.
13. Xia J., Yin B., Liu L., Lu Y., Geng D., Tian W. Imaging features of pilocytic astrocytoma in cerebral ventricles. Clin. Neuroradiol. 2016 Sep. 26(3). 341-6. doi: 10.1007/s00062-015-0370-6.
14. Roskams T. The role of immunohistochemistry in diagnosis. Clin. Liver Dis. 2002 May. 6(2). 571-89, x. doi: 10.1016/s1089-3261(02)00012-0.
15. Kristensen B.W., Priesterbach-Ackley L.P., Petersen J.K., Wesseling P. Molecular pathology of tumors of the central nervous system. Ann. Oncol. 2019 Aug 1. 30(8). 1265-1278. doi: 10.1093/annonc/mdz164.
16. Tian Z., Chen C., Fan Y., Ou X., Wang J., Ma X., Xu J. Glioblastoma and anaplastic astrocytoma: differentiation using MRI texture analysis. Front. Oncol. 2019. 9. 876. doi: 10.3389/fonc.2019.00876.
17. Alshoabi S.A., Alareqi A.A., Omer A.M., Suliman A.G., Daqqaq T.S. Diffuse astrocytoma and the diagnostic dilemma of an unusual phenotype: a case report. Radiol. Case Rep. 2020 Dec 3. 16(2). 319-326. doi: 10.1016/j.radcr.2020.11.023.
18. Mubarak F., Naeem A. Imaging and histopathological features of pilocytic astrocytoma involving various locations of central nervous system: series of multiple cases. Journal of Neurophysiology and Neurological Disorders. 2021. 9. 1-7.
19. Barker C.A., Chang M., Beal K., Chan T.A. Survival of patients treated with radiation therapy for anaplastic astrocytoma. Radiol. Oncol. 2014 Nov 5. 48(4). 381-6. doi: 10.2478/raon-2014-0019.
20. Forst D.A., Nahed B.V., Loeffler J.S., Batchelor T.T. Low-grade gliomas. Oncologist. 2014 Apr. 19(4). 403-13. doi: 10.1634/theoncologist.2013-0345.
21. Pace A., Villani V., Benincasa D., Di Pasquale A., Carapella C.M., Pompili A. P18.07: palliative sedation for brain tumor patients at the end of life. Neuro Oncol. 2014 Sep. 16(Suppl. 2). ii1-ii112. doi: 10.1093/neuonc/nou174.431.
22. Walbert T., Chasteen K. Palliative and supportive care for glioma patients. Cancer Treat. Res. 2015. 163. 171-84. doi: 10.1007/978-3-319-12048-5_11.
23. Narita Y., Chernov M.F., Muragaki Y., Kesari S., McCut-cheon I.E. Chemotherapy of diffuse astrocytoma (WHO grade II) in adults. Intracranial gliomas. Part II: Adjuvant therapy. Programming Neurology Surgery. 2018. 31. 145-151. doi: 10.1159/000467375.
24. Wang G.W., Li B.M. Efficacy of surgery for the treatment of astrocytoma: a protocol of systematic review and meta-analysis. Medicine (Baltimore). 2020 Jun 5. 99(23). e20485. doi: 10.1097/MD.0000000000020485.
25. Wahner H.C.W., Träger M., Bender K. et al. Predicting survival in anaplastic astrocytoma patients in a single-center cohort of 108 patients. Radiat. Oncol. 2020. 15. 282. doi: 10.1186/s13014-020-01728-8.
26. Steidl E., Filipski K., Zeiner P.S., Wagner M., Fokas E., Forster M.T., Ronellenfitsch M.W., Divé I., Steinbach J.P., Harter P.N., Bähr O. A 25-year retrospective, single center analysis of 343 WHO grade II/III glioma patients: implications for grading and temozolomide therapy. J. Cancer Res. Clin. Oncol. 2021 Aug. 147(8). 2373-2383. doi: 10.1007/s00432-021-03511-y.
27. Hervey-Jumper S.L., Berger M.S. Role of surgical resection in low- and high-grade gliomas. Curr. Treat. Options Neurol. 2014 Apr. 16(4). 284. doi: 10.1007/s11940-014-0284-7.
28. Xia L., Fang C., Chen G., Sun C. Relationship between the extent of resection and the survival of patients with low-grade gliomas: a systematic review and meta-analysis. BMC Cancer. 2018 Jan 6. 18(1). 48. doi: 10.1186/s12885-017-3909-x.
29. Patel S.H., Bansal A.G., Young E.B., Batchala P.P., Patrie J.T., Lopes M.B., Jain R., Fadul C.E., Schiff D. Extent of surgical resection in lower-grade gliomas: differential impact based on molecular subtype. AJNR. 2019 Jul. 40(7). 1149-1155. doi: 10.3174/ajnr.A6102.
30. Wang L., Liang B., Li Y.I. et al. What is the advance of extent of resection in glioblastoma surgical treatment — a systematic review. Chin. Neurosurg. J. 2019. 5. 2. doi: 10.1186/s41016-018-0150-7.
31. Choi J., Kim S.H., Ahn S.S. et al. Extent of resection and molecular pathologic subtype are potent prognostic factors of adult WHO grade II glioma. Sci. Rep. 2020. 10. 2086. doi: 10.1038/s41598-020-59089-x.
32. Laurent D., Freedman R., Cope L., Sacks P., Abbatematteo J., Kubilis P., Bova F., Rahman M. Impact of extent of resection on incidence of postoperative complications in patients with glioblastoma. Neurosurgery. 2020 May 1. 86(5). 625-630. doi: 10.1093/neuros/nyz313.
33. Mao H., Li X., Mao W. Advantages of gross total resection in patients with astrocytoma: a population-based study. Oncol. Lett. 2020 Jun. 19(6). 3761-3774. doi: 10.3892/ol.2020.11514.
34. Karschnia P., Vogelbaum M.A., van den Bent M., Cahill D.P., Bello L., Narita Y., Berger M.S., Weller M., Tonn J.C. Evidence-based recommendations on categories for extent of resection in diffuse glioma. Eur. J. Cancer. 2021 May. 149. 23-33. doi: 10.1016/j.ejca.2021.03.002.
35. Motomura K., Chalise L., Ohka F., Aoki K., Tanahashi K., Hirano M., Nishikawa T., Yamaguchi J., Shimizu H., Wakabayashi ., Saito R. Impact of the extent of resection on the survival of patients with grade II and III gliomas using awake brain mapping. J. Neurooncol. 2021 Jun. 153(2). 361-372. doi: 10.1007/s11060-021-03776-w.
36. Revilla-Pacheco F., Rodríguez-Salgado P., Barrera-Ramírez M., Morales-Ruiz M.P., Loyo-Varela M., Rubalcava-Ortega J., Herrada-Pineda T. Extent of resection and survival in patients with glioblastoma multiforme: systematic review and meta-analysis. Medicine (Baltimore). 2021 Jun 25. 100(25). e26432. doi: 10.1097/MD.0000000000026432.

/24.jpg)
/25.jpg)
/27.jpg)