Definition
Meningiomas of the skull base constitute 40 % of all intracranial meningiomas. Of these, about one-half occur in the sphenoid wing. Anatomically, the sphenoid wing meningioma (SWM) refers to tumors that originate in any part of the bony crest formed by wings (lesser and greater) of the sphenoid bone; it is the boundary between the anterior and middle cranial floor. The lesser wing constitutes its internal two-thirds and the most complex area, while the greater wing forms its external third [5–7].
Molecular biology and clinicopathologic features
Meningiomas were among the first tumors analyzed for genetic abnormalities. Neurofibromatosis type 2 is the most well-known familial syndrome, which is caused by the germline mutation of the gene on 22q12.2. Over 50 % of individuals in this syndrome manifests at least one meningioma in their lifetime, with a mean age of 30 years. Most of the meningiomas in NF2 disorder background present a fibrous or transitional phenotype and are generally more aggressive than sporadic tumors. Losses on 1p and 14q are the next most common mutation after LOH 22q, and these aberrations are independently correlated with increased tumor grade and recurrence rate [8–10].
Recently, several seminal studies evaluating the geno-mics of meningiomas have rapidly changed the understan-ding of the disease. One of the landmark early study by Clark et al. identified a series of mutations in non-NF2-mutated meningiomas, namely, tumor necrosis factor, receptor-associated factor 7 (TRAF7), Kruppel-like factor 4, AKT1, Smoothened (SMO), and PIK3CA. An advantage of this study is the understanding of these single genetic changes on a broad scale and their implications on the clinical behavior and progression of meningiomas [11].
The difference in the embryonic origins of the meninges according to site is associated with the histology, location, and recurrent mutations of meningiomas. Meninges of the skull base originate from mesoderm whereas meninges of the convexity originate from the neural crest. Meningothelial variants are more frequently found in the skull base with 15–25% frequency of gene TRAF7, with early tumor recurrence prognosis; whereas fibrous meningiomas mainly develop in the convexity. Moreover, genetic alterations of the NF2 gene are preferentially found in the convexity, and most other genetic alterations except NF2 are mainly found in skull base tumors. The tumor site may also be related to the histologic grade, as the proportion of grade II and III tumors are much higher at the convexity than at the skull base, where grade I is more common at the skull base [9].
Other study found that oncogenic of SMO and AKT1 mutations are likely to be high yield in both WHO grade I and II of anterior skull base and noted that patients with AKT1-mutated meningiomas presented at a younger age of 45.0 ± 8.5 years, where SMO mutations presented with older age and larger tumor [12].
Classification of SWM and diagnosis
In 1938, Cushing and Eisenhardt classified SWMs into two main varieties: en plaque and globoid.
En plaque meningiomas
Also known as spheno-orbital meningiomas or hyperostotic meningiomas, it is referred to as tumors with carpet-like dural growth, which are associated with a reactive hyperostosis [13, 14]. Sphenoid wing hyperostosis has been reported in as many as 42 % of all meningiomas in this area and should be differentiated with fibrous dysplasia, osteoma, osteoblastic metastasis [14–17]. The primary clinical manifestations of these lesions are proptosis and oculomotor deficits [13]. Hyperostosis in meningiomas was initially described by Brissaud and Lereboullet in 1903 [18]. The difference in theories regarding the cause of hyperostosis includes vascular disturbances, irritation of bone without actual invasion, previous trauma, bone production by tumor cells, or osteoblastic stimulation of normal bone. Currently, the most widely accepted theory is bone invasion by tumor [4, 18].
Globoid meningiomas
They are classified into three groups: 1) inner, or clinoidal, 2) middle or alar, and 3) lateral, outer, or pterional.
Middle meningiomas have radiologic characteristics similar to lateral meningiomas and surgical resection and clinical results of both types are almost identical. For this reason, some authors suggest that globoid meningiomas of the sphenoid wing can be classified into only two groups: deep, inner, or clinoidal and lateral, outer, or pterional, discharging the middle or alar variety [19].
Based on intraoperative anatomical observations, O. Al-Mefty classified three subgroups of clinoidal meningiomas (CM): group I, lower CM, no arachnoidal dissection plane between the internal carotid artery (ICA) and tumor; group II, distal or lateral CM, an arachnoidal membrane of the carotid cistern separates the tumor from the adventitia making dissection possible; group III, CM that originates in the optic foramen, the arachnoidal membrane is present between the ICA and the tumor but may be absent between the optic nerve and the tumor. Meningiomas are characterized by severe visual loss with optic atrophy on one side [5].
There is a third and debatable variety of SWMs. These are tumors that grow within the diploë without an epidural, or subcutaneous component and are referred to as intraosseous or intradiploic meningiomas [4, 14]. The origin of these tumors in the skull base is controversial. Arachnoid cells have been described, following the vessels and nerves in bone foramina or trapped within the sutures. However, some authors [20] doubt the existence of these tumors, proposing that they are really a variety of en plaque meningiomas.
Symptoms
The SWM can grow in any direction and cause neurological deficit based on the adjacent structures by the compression or encasement. Medial compression or invasion of the cavernous sinus, compression of the ICA and cranial nerves III, IV, V, and VI may cause double vision. Superiorly, the ICA and its branches outside the cavernous sinus can be encased causing transient ischemic attack or stroke.
The SWM grows anteriorly through the optic canal
and/or above it to the anterior fossa leading to reduced visual acuity and/or causing frontal lobe compression with potential cognitive impairment. Laterally compression of the temporal lobe causes seizure and memory impairment. Posterior compression of the oculomotor nerve and the midbrain in the interpeduncular cistern causes double vision (Fig. 1A, B) [21].
Radiological diagnosis
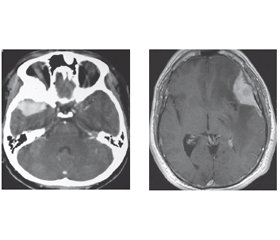
In neurosurgical practice, the radiological diagnosis of meningioma is typically certain on computed tomography (CT) and magnetic resonance imaging (MRI) evaluation (Fig. 2). However, differentiating residual or recurrent tumors from postsurgical or radiation treatment changes may be a challenge. Recently modern imaging modalities such as nuclear scintigraphy or positron emission tomography have the benefit of meningioma biology. An evidence-based recommendation for the use of molecular imaging in meningioma has been proposed recently. Conventional angio-graphy is indicated in the diagnostic work-up, only when the diagnosis is in question and most often in association with pre-operative endovascular embolization to minimize blood loss intraoperatively [22].
Treatment
Treatment of meningiomas depends on the tumor size, tumor location, associated symptoms, age, and health status.
Asymptomatic meningioma: the routine use of a CT and MRI in neurosurgical practice, for investigation of different pathologies, results in the discovery of unexpected incidental (asymptomatic), incidentally discovered meningiomas (IDMs). Approximately 32 % of IDMs do not grow within 3 years of the follow-up period. Data are lacking to make evidence-based management guidelines. A suggestion is to obtain a follow-up imaging study 3–4 months after the initial study, to rule out rapid progression and then repeat annually [23]. Observational studies of meningiomas, with average follow-up periods from 2.8 to 7.8 years have found that 20 to 40 % of asymptomatic meningiomas grow at a li-near rate of 2 to 3 mm per year. However, volumetric analysis is the most accurate way to estimate a growth rate [24].
O. Al-Mefty advocate the “wait-and-see” approach in IDMs, with exception of the tuberculum meningiomas, because of the possibility that future small growth could end up rapidly affecting vision irreparably. This opinion is based on the majority of studies showing that the natural history of IDMs is generally benign and indolent. This recommendation is definitely applied in older individuals, in those who are medically unstable, or in those who do not desire surgery, and radiosurgery is not primary treatment when progression has not been documented [25]. Other reviews demonstrated that high T2-signal and non-calcification were associated with symptom development and intervention, respectively, which may have been due to meningioma growth [26].
Surgical treatment
Surgical treatment is, without doubt, the best option for SWMs. This treatment is indicated based on the size of the lesion, presence of signs or symptoms, patient’s condition, changes of the edema on imaging studies, and surgeon’s experience. In general, surgery is indicated to all patients who are in good health and have a tumor size greater than 2.5 cm. For smaller tumors that are not too close to the visual system, stereotactic radiosurgery (SRS) might be considered. The goal of the surgery in all cases should be radical excision of the tumor, which means resection of the lesion, along with the dural implant (1-cm margin) and all hyperostotic bone, except in some spheno-orbital and clinoidal meningiomas, and invasion to the cavernous sinus. For these cases, most authors recommend excising the tumor, dura mater, and infiltrated bone on extracavernous areas but leaving the intracavernous portion for another adjuvant treatment, such as radiosurgery, because even in experienced hands, the third nerve morbidity is extremely high after a direct approach to this region [9, 27–29].
Radiation
The first clinical applications of SRS for meningioma were performed by Lars Leksell in 1970 [30]. The stereotactic radiosurgery and stereotactic radiotherapy (SRT) have been successfully applied when secondary surgical resection was not practical or feasible [31]. The SRS typically delivers a highly conformal, high dose of radiation in a single fraction and has been applied more frequently to the practice. It is used after SRT or at the recurrence and as a definitive primary treatment for presumed benign meningiomas. The SRS is usually considered effective and safe for a tumor that is limited in size up to 3 cm in maximal diameter or 10 cc in volume and with sufficient distance from critical structures such as the optic chiasm, optic nerve, or brainstem [32]. The median dose for SRS was 15 Gy and the usual regimen was 25 Gy in five fractions for hypofractionated SRT [33]. Stereotactic radiosurgery has a higher risk of clinical deficits and symptomatic edema in comparison with SRT. However, progression-free survival is not significantly different across the two techniques. Hypofractionated stereotactic radiosurgery may be an option for patients with larger skull base meningioma [34].
Chemotherapeutic agents
Chemotherapeutic agents have been investigated and most have failed to show consistent efficacy to patients who have exhausted all surgical and radiotherapy options. Most commonly used agents in the treatment of meningioma include hydroxyurea [35–37], somatostatin analogues [38], and hormonal agents such as progesterone receptor inhibitors. Interferon-alpha and somatostatin analogues are among the biological agents showing responses in patients with recurrent meningioma [39].
Recurrenuce and outcomes
In addition to the extent of surgical resection, the WHO grading of meningiomas is another major predictive factor of recurrence. For WHO grade I with gross total resection, the recurrence rate ranges from 7 to 25 % [40–42]. Although some histologic variants of benign (grade I) meningioma like secretory are associated with increased findings of peritumoral brain edema on preoperative imaging, no differential growth or recurrence rate has been identified between histologic subgroups of benign meningiomas [41]. Atypical meningiomas WHO grade II are associated with recurrence rates of 29 to 52 %, and anaplastic meningiomas WHO grade III recur in 50 to 94 % of cases [42].
According to the results of our own study, among 67 patients with SWM within mean follow-up of 87 months, recurrence was in 16.4 % of cases and was strongly related to pathological grade (grade II, atypical type) and Simpson grade [43]. Recent studies have questioned the relevance of the extent of resection for recurrence in the era of modern imaging and microsurgical technique. They showed no significant diffe-rence in recurrence-free survival between patients receiving various Simpson grade resections [44]. These results were disputed by others who showed that the extent of resection continues to be a significant factor in predicting tumor recurrence [45, 46]. Other researchers found that the extent of resection is not equally prognostic for all tumor locations, with convexity meningiomas recurrence rates being more related to the extent of resection than parasagittal and falcine meningiomas [47]. Other prognostic factors that have been shown to impact the recurrence include the degree of histologic atypia and MIB-1 index [48, 49]. Moreover, younger age and the absence of calcification, along with higher MRI T2 signal intensity, are associated with a faster rate of growth of incompletely resected meningiomas [50].
Conclusions
Surgical resection is clearly the standard in the meningiomas management for symptomatic patients; it is more likely to relieve symptoms and mass effect than radiation alone. Advances in molecular techniques and optimum understanding of anatomical and radiological fields provide more favorable results and outcomes.
Received 27.01.2022
Revised 10.02.2022
Accepted 15.02.2022
Список литературы
1. MacCarty C.S. Meningiomas of the sphenoidal ridge. J. Neurosurg. 1972 Jan. 36 (1). 114-20. doi: 10.3171/jns.1972.36.1.0114. PMID: 5007268.
2. Guduk M., Ozduman K., Pamir M. Necmettin. Sphenoid Wing Meningiomas: Surgical Outcomes in a Series of 141 Cases and Proposal of a Scoring System Predicting Extent of Resection. World Neurosurg. 2019. doi: 10.1016/j.wneu.2018.12.175. PMID: 30639480.
3. Pieper D.R., Al-Mefty O., Hanada Y., Buechner D. Hyperostosis associated with meningioma of the cranial base: secondary changes or tumor invasion. Neurosurgery. 1999. 44. 742-746. doi: 10.1097/00006123-199904000-00028. PMID: 10201298.
4. Guinto G., Abello J., Félix I. et al. Lesions confined to the sphenoid ridge. Differential diagnosis and surgical treatment. Skull Base Surg. 1997. 7. 115-121.
5. Al-Mefty O. Clinoidal meningiomas. J. Neurosurg. 1990. 73 (6). 840-9. doi: 10.3171/jns.1990.73.6.0840.
6. Ojemann R.G. Meningiomas: clinical features and surgical management. In: Wilkins R.H., Rengachary S.S., eds. Neurosurgery. New York: McGraw-Hill, 1985. 635-654.
7. Brotchi J., Pirotte B. Sphenoid wing meningiomas. In: Sekhar L.N., Fessler R.G., eds. Atlas of Neurosurgical Techniques. Brain. New York: Thieme, 2006. 623-632.
8. DeMonte F., McDermott M.W., Al-Mefty O. Meningiomas. Second edition. New York: Thieme, 2011. P. 4-5.
9. Lee Y.S., Lee Y.S. Molecular characteristics of meningiomas. J. Pathol. Transl. Med. 2020 Jan. 54(1). 45-63. doi: 10.4132/jptm.2019.11.05. Epub 2020 Jan 15. PMID: 31964111. PMCID: PMC6986967.
10. Adrian Maurer, Jacob Archer, Sam Safavi-Abbasi, Michael Sughrue. A review of the histologic, genetic and molecular characteristics of meningioma pathogenesis and progression. J. of Cancer Genetics and Biomarkers. 2013. 1(2). 24-38. doi.org/10.14302/issn.2572-3030.jcgb-14-383
11. Michael Karsy, Mohammed A. Azab, Hussam Abou-Al-Shaar, Jian Guan, Ilyas Eli, Randy L. Jensen, D. Ryan Ormond. Clinical potential of meningioma genomic insights: a practical review for neurosurgeons. Neurosurg. Focus. 2018. 44(6). E10. doi: 10.3171/2018.2.FOCUS1849. PMID: 29852774.
12. Matthew R. Strickland, Corey M. Gill, Naema Nayyar, Megan R. D’Andrea, Christian Thiede, Tareq A. Juratli et al. Targeted sequencing of SMO and AKT1 in anterior skull base meningiomas. J. Neurosurg. 2017. 127. 438-444.
13. Ouyang T., Zhang N., Wang L., Li Z., Chen J. Sphenoid wing meningiomas: surgical strategies and evaluation of prognostic factors incluencing clinical outcomes. Clin. Neurol. Neurosurg. 2015. 134. 85-90. doi: 10.1016/j.clineuro.2015.04.016.
14. Simas N.M., Farias J.P. Sphenoid wing en plaque meningiomas: surgical results and recurrence rates. Surg. Neurol. Int. 2013. 4. 86.
15. Bassiouni H., Asgari S., Sandalcioglu E. et al. Anterior clinoidal meningiomas: functional outcome after microsurgical resection in a consecutive series of 106 patients. J. Neurosurg. 2009. 111. 1078-1090.
16. Goel A., Gupta S., Desai K. New grading system to predict resectability of anterior clinoid meningiomas. Neurol. Med. Chir. (Tokyo). 2000. 40. 610-617. doi: 10.2176/nmc.40.610. PMID: 11153190.
17. Honeybul S., Neil-Dwyer G., Lang D.A. et al. Sphenoid wing meningiomas en plaque: a clinical review. Acta Neurochir. (Wien). 2001. 143. 749-758.
18. Bikmaz K., Mrak R., Al-Mefty O. Management of bone-invasive, hyperostotic sphenoid wing meningiomas. J. Neurosurg. 2007. 107. 905-912.
19. Russell S.M., Benjamin V. Medial sphenoid ridge meningiomas: classification, microsurgical anatomy, operative nuances, and long-term surgical outcome in 35 consecutive patients. Neurosurgery. 2008. 62 (3, suppl. 1). 38-50.
20. Ringel F., Cedzich C., Schramm J. Microsurgical technique and results of a series of 63 spheno-orbital meningiomas. Neurosurgery. 2007. 60 (4, suppl. 2). ONS214-ONS222.
21. Schmidek & Sweet. Operative neurosurgical techniques: indications, methods, and results. Philadelphia: Elsevier, 2022. Seventh ed., chap. 7. P. 309-315.
22. Raymond Y. Huang, Wenya Linda Bi, Brent Griffith, Timothy J. Kaufmann, Christian la Fougère, Nils Ole Schmidt, Jöerg C. Tonn, Michael A. Vogelbaum, Patrick Y. Wen, Kenneth Aldape, Farshad Nassiri, Gelareh Zadeh, Ian F. Dunn, The international consortium on meningiomas. Imaging and diagnostic advances for intracranial meningiomas. Neuro Oncology. 2019. 21(s1). 44-61.
23. Mark S. Greenberg. Handbook of Neurosurgery. New York: Thieme, 2019. Ninth Edition. P. 712.
24. Yano S., Kuratsu J. Kumamoto; Brain Tumour Research Group. Indications for surgery in patients with asymptomatic meningiomas based on an extensive experience. J. Neurosurg. 2006. 105(4). 538-543.
25. Al-Mefty O. Controversies in neurosurgery II. New York: Thieme, 2014. P. 56.
26. Abdurrahman I. Islim, Midhun Mohan, Richard D.C., Moon Nisaharan Srikandarajah, Samantha J. Mills, Andrew R. Brodbelt, Michael D. Jenkinson. Incidental intracranial meningiomas: a syste-matic review and metaanalysis of prognostic factors and outcomes. Journal of Neurooncology. 2019. 142. 211-221. doi: 10.1007/s11060-019-03104-3. PMID: 30656531.
27. Basso A., Carrizo A.G., Duma C. Sphenoid ridge meningiomas. In: Schmidek H.H., ed. Operative Neurosurgical Techniques. Indications. Methods and Results. Philadelphia: WB Saunders, 2000. 316-324.
28. Nakamura M., Roser F., Vorkapic P. et al. Medial sphenoid wing meningiomas: clinical outcome and recurrent rate. Neurosurgery. 2006. 58. 626-639.
29. Yang J., Ma S.C., Liu Y.H. et al. Large and giant medial sphenoid wing meningiomas involving vascular structures: clinical features and management experience in 53 cases. Clin. Med. J. (Engl). 2013. 126. 4470-4476.
30. Leksell L. Stereotactic radiosurgery. J. Neurol. Neurosurg. Psychiatry. 1983 Sep. 46(9). 797-803. doi: 10.1136/jnnp.46.9.797. PMID: 6352865.
31. Kim M., Cho Y.H., Kim J.H., Kim C.J., Kwon D.H. Analysis the causes of radiosurgical failure in intracranial meningiomas treated with radiosurgery. Clin. Neurol. Neurosurg. 2017. 154. 51-58.
32. Fabio Y. Moraes, Caroline Chung. Radiation for skull base meningiomas: review of the literature on the approach to radiotherapy. Chin. Clin. Oncol. 2017. 6 (Suppl. 1). S3.
33. Unger K.R., Lominska C.E., Chanyasulkit J., Randolph-Jackson P., White R.L., Aulisi E., Jacobson J., Jean W., Gagnon G.J. Risk factors for posttreatment edema in patients treated with stereotactic radiosurgery for meningiomas. Neurosurgery. 2012. 70. 639-45.
34. Nida Fatima, Antonio Meola, Erqi L. Pollom, Scott G. Soltys, Steven D. Chang. Stereotactic radiosurgery versus stereotactic radiotherapy in the management of intracranial meningiomas: a systematic review and meta-analysis. Neurosurg. Focus. 2019. 46(6). E2.
35. Wen P.Y., Quant E., Drappatz J., Beroukhim R., Norden A.D. Medical therapies for meningiomas. J. Neurooncol. 2010. 99. 365-378.
36. Loven D., Hardoff R., Sever Z.B. et al. Non-resectable slow-growing meningiomas treated by hydroxyurea. J. Neurooncol. 2004. 67. 221-226.
37. Newton H.B., Scott S.R., Volpi C. Hydroxyurea chemotherapy for meningiomas: enlarged cohort with extended follow-up. Br. J. Neurosurg. 2004. 18. 495-499.
38. Chamberlain M.C., Glantz M.J., Fadul C.E. Recurrent meningioma: salvage therapy with long-acting somatostatin analogue. Neurology. 2007. 69. 969-973.
39. Kaba S.E., DeMonte F., Bruner J.M. et al. The treatment of recurrent unresectable and malignant meningiomas with interferon alpha-2B. Neurosurgery. 1997. 40. 271-275.
40. Perry A., Stafford S.L., Scheithauer B.W., Suman V.J., Lohse C.M. Meningioma grading: an analysis of histologic parameters. Am. J. Surg. Pathol. 1997. 21. 1455-1465.
41. Perry A., Louis D., Budka H. et al. Meningiomas. In: Louis D., Ohgaki H., Wiestler O., Cavenee W., eds. WHO Classif. Tumours Cent. Nerv. Syst. Geneva: International Agency for Research on Cancer, 2016. 232-237.
42. Lamszus K. Meningioma pathology, genetics, and biology. J. Neuropathol. Exp. Neurol. 2004. 63. 275-286.
43. Abdalrahman M. Nassar, Volodymyr I. Smolanka, Andriy V. Smolanka, Evelina Z. Murzho, Dipak Chaulagain. Recurrence rate of sphenoid wing meningiomas and role of peritumoural brain edema: a single center retrospective study. UNJ. 2021. Vol. 27. N 4. 38-45. doi: 10.25305/unj.242064.
44. Sughrue M.E., Kane A.J., Shangari G. et al. The relevance of Simpson Grade I and II resection in modern neurosurgical treatment of World Health Organization Grade I meningiomas. J. Neurosurg. 2010. 113. 1029-1035. doi: 10.3171/2010.3.JNS091971.
45. Hasseleid B.F., Meling T.R., Rønning P., Scheie D., Hel-seth E. Surgery for convexity meningioma: Simpson Grade I resection as the goal: clinical article. J. Neurosurg. 2012. 117. 999-1006. doi: 10.3171/2012.9.JNS12294.
46. Nanda A., Bir S.C., Konar S., Maiti T.K., Bollam P. World Health Organization grade I convexity meningiomas: study on outcomes, complications and recurrence rates. World Neurosurg. 2016. 89. 620-627. doi: 10.1016/j.wneu.2015.11.050. e2.
47. Voß K.M., Spille D.C., Sauerland C. et al. The Simpson grading in meningioma surgery: does the tumor location influence the prognostic value? J. Neurooncol. 2017. 133. 641-651. doi: 10.1007/s11060-017-2481-1.
48. Marciscano A.E., Stemmer-Rachamimov A.O., Niemierko A. et al. Benign meningiomas (WHO Grade I) with atypical histological features: correlation of histopathological features with clinical outcomes. J. Neurosurg. 2016. 124. 106-114. doi: 10.31 71/2015.1.JNS142228.
49. Oya S., Kawai K., Nakatomi H., Saito N. Significance of Simpson grading system in modern meningioma surgery: integration of the grade with MIB-1 labeling index as a key to predict the recurrence of WHO Grade I meningiomas. J. Neurosurg. 2012. 117. 121-128. doi: 10.3171/2012.3.JNS111945.
50. Nakamura M., Roser F., Michel J., Jacobs C., Samii M. Volumetric analysis of the growth rate of incompletely resected intracranial meningiomas. Zentralbl. Neurochir. 2005. 66. 17-23. doi: 10.1055/s-2004-836225.

/44.jpg)
/45.jpg)