Introduction
Oligoastrocytoma (OA), an infiltrating glioma with oligodendroglial and astrocytic features, has been traditionally defined as an oligodendroglial and astrocytic tumor [1]. OA diagnosis should be avoided based on the fourth edition of the World Health Organization classification of tumours of the central nervous system since molecular testing may distinguish between the two types of glioma [2]. OA was categorized as a separate entity within diffuse gliomas. Glioblastoma multiforme was classified in the literature as “a diffusely infiltrating tumor consisting of two separate tumor cell types that have certain characteristics with those of oligodendrogliomas or tumors with diffuse angiogenesis” [3]. Research has shown that almost all OAs are autologous tumors that may be categorized as either astrocytomas (the most common OA) or oligodendrogliomas (the second most common type) [4–7].
Given that assessing overall survival may be challenging because the majority of patients have prolonged survival duration, the primary target of any treatment should be to preserve the patient’s neurological functioning and quality of life. Several studies have shown that surgical excision of low-grade glioma (LGG) has a considerable effect on the tumor’s natural history [8–11]. Unfortunately, a significant percentage of LGGs are located in highly expressive brain areas, making effective removal impossible. Additionally, in case of a diffusive pattern of growth, surgery offers relatively little oncologically useful resection area. After a biopsy has been conducted in this group of individuals, radiation or chemotherapy may be used to help them recover.
We provide a case of a patient who had partial resection of an oligoastrocytoma using functional magnetic resonance imaging (fMRI) and intraoperative neuromonitoring to ensure maximal resection while preserving the motor and speech centers.
Case report
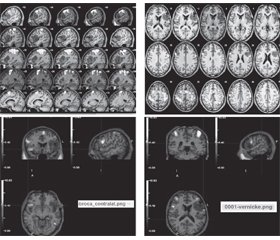
It has been a year since a 30-year-old male with no neurological condition began experiencing a partial seizure affecting the left arm and lower leg. On T1-weighted magnetic resonance images, large left fronto-parieto-temporal tumors were discovered in the Broca’s and Wernicke’s areas. The location of the Broca’s and Wernicke’s areas was determined using functional magnetic resonance imaging. The fMRI technique was used to better understand the position of the right hand’s motor center in relation to its anatomical location in the left hemisphere (Fig. 1).
/24.jpg)
The patient was advised to have a craniotomy as a part of our surgical resection strategy. Craniotomy was performed with the support of intraoperative neuromonitoring in order to accomplish maximal resection with minimal harm to the eloquent region. When comparing pre- and post-surgical volumes, a total of 63.5 % of the tumor was removed. According to volumetric analysis using T1 MRI performed before surgery and 72 hours after it, post-operative tumor volume was 92.51 cm3 and pre-operative — 253.73 cm3 (Fig. 2).
Histological examination revealed that the MGMT promoter was unmethylated, which matched the preceding findings with a somewhat higher labeling index for Ki-67 of 4 %. Three months after the patient’s discharge from the hospital where he was recommended chemotherapy, a follow-up examination found that the tumor had stabilized but the frequency of seizures had decreased just one time in that period and there were no signs of hemiparesis.
Discussion
Oligoastrocytoma is a subtype of glial cell continuum that also includes astrocytic and oligodendrocytic cells. The World Health Organization classifies these cancers as low-grade or anaplastic. Increased cell density, pleomorphic, nuclear atypia and higher mitotic activity are all hallmarks of an anaplastic oligoastrocytoma. Diagnosis does not need the presence of microvascular proliferation and necrosis [12].
Young patients (mean diagnosis age 35–45 years) often have partial or generalized seizures as a symptom of these malignancies [13]. Although less common, individuals may present with headaches or have no symptoms at all [14]. Most oligoastrocytomas are found in the frontal or temporal lobes, according to Beckmann and Prayson [15]. If an intracranial mass is suspected, patients often undergo non-enhanced computed tomography (CT) of the head as the first step in diagnostic evaluation. Oligoastrocytomas are commonly seen on CT images as intraaxial low-attenuation regions without concomitant edema. Unlike similar high-grade lesions, these tumors are less likely to have a significant influence on the population as a whole because of their slow growth. T1-weighted images show a hypointense lesion, whereas T2-weighted images show a hyperintense lesion. Contrast material enhancement may be seen in around half of the oligoastrocytoma patients examined by Shaw et al. [17].
Low-grade oligoastrocytoma survival has not improved much despite advances in diagnosis and the appearance of more intensive treatment regimens. For patients with low-grade glioma, a recent research demonstrated a statistically significant association between resection and overall survival (both total and absence of cancer development) [11]. Surgical intervention remains the initial step, with the objective of total resection [18]. A postoperative MRI must be performed to ensure that the surgical resection was as complete as feasible. Surgical excision is ineffective for infiltrative tumors in critical locations, even if such criteria are applied to large lesions in non-eloquent areas. Since these tumors are often treated with radiotherapy, radiation-induced damage must still be taken into consideration when determining treatment choices [19]. Even while no agreement has been achieved on the regular use of chemotherapy in the treatment of LGG, especially astrocytomas, its effectiveness in terms of clinical and radiographic response has been proven either as an initial treatment [20–23] or post-surgical and radiation treatment [24, 25]. A successful response is defined as volumetric shrinking or stablity over time, while full neuroradiological remission is very rare. To understand why chemotherapy response varies amongst histologically similar cancers, it is necessary to understand how genetic aberrations accumulate throughout progression from low- to high-grade malignancies [26].
The tumor had been partly resected in our patient, according to postoperative MRI results. It was not possible to remove the entire tumor during surgery, but it did reduce the size and effect of the tumor, as well as the frequency of the patient’s seizures. If you want to know how a treatment affects overall survival, you’ll need long-term monitoring.
Conclusions
To treat low-grade oligoastrocytoma, surgical resection and subsequent chemotherapy are critical. In certain patients, a whole or partial resection may considerably reduce tumor volume, allowing for future therapy. As a consequence, this combination may delay radiation and improve quality of life. More studies are needed to identify people who respond well to treatment.
Received 05.01.2022
Revised 09.01.2022
Accepted 15.01.2022
Список литературы
1. Louis D.N., Ohgaki H., Wiestler O.D., Cavenee W.K., Bur-ger P.C., Jouvet A., Scheithauer B.W., Kleihues P. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007. 114. 97-109.
2. Louis D.N., Perry A., Reifenberger G., von Deimling A., Fi-garella-Branger D., Cavenee W.K., Ohgaki H., Wiestler O.D., Kleihues P., Ellison D.W. The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol. 2016. 131. 803-820.
3. Von Deimling A., Refeinberger G., Kros J.M. et al. Oligoastrocytoma. In: Luois D.N., Oghaki H., Wiestler O.D., Cavenee W.K., eds. WHO classification of tumors of the central nervous system. Lyon: IARC Press; 2007. 63-66.
4. Jiao Y., Killela P.J., Reitman Z.J. et al. Frequent ATRX, CIC, FUBP1 and IDH1 mutations refine the classification of malignant gliomas. Oncotarget. 2012. 3. 709-22.
5. Sahm F., Reuss D., Koelsche C. et al. Farewell to oligoastrocytoma: in situ molecular genetics favor classification as either oligodendroglioma or astrocytoma. Acta Neuropathol. 2014. 128. 551-9.
6. Wiestler B., Capper D., Sill M. et al. Integrated DNA methylation and copy-number profiling identify three clinically and biologically relevant groups of anaplastic glioma. Acta Neuropathol. 2014. 128. 561-71.
7. Hewer E., Vajtai I., Dettmer M.S. et al. Combined ATRX/IDH1 immunohistochemistry predicts genotype of oligoastrocytomas. Histopathology. 2016. 68. 272-8.
8. Berger M.S., Deliganis A.V., Dobbins J.D., Keles G.E. The effect of extent of resection on recurrence in patients with low-grade cerebral hemisphere gliomas. Cancer. 1994. 74. 1748-1791.
9. Duffau H., Lopes M., Arthuis F., Bitar A., Sichez J.P., Van Effenterre R., Capelle L. Contribution of intraoperative electrical stimulations in surgery of low grade gliomas: a comparative study between two series without (1985–1996) and with (1996–2003) functional mapping in the same institution. J. Neurol. Neurosurg. Psychiatry. 2005. 76. 845-851.
10. Keles G.E., Lamborn K.R., Berger M.S. Low grade hemispheric gliomas in adults: a critical review of extent of resection as a factor influencing outcome. J. Neurosurg. 2001. 95. 735-745.
11. Smith J.S., Chang E.F., Lamborn K.R., Chang S.M., Prados M.D., Cha S., Tihan T., Vandenberg S., McDermott M.W., Berger M.S. Role of extent of resection in the long-term outcome of low grade hemispheric gliomas. J. Clin. Oncol. 2008. 26. 1338-1345.
12. Reifenberger G., Kros J.M., Burger P.C., Louis D.N., Collins V.P. Oligoastrocytoma. In: Kleihues P., Cavenee W.K., eds. Tumours of the nervous system. Lyon, France: IARC Press; 2000. 65-67.
13. Behin A., Hoang-Xuan K., Carpentier A.F., Delattre J.Y. Primary brain tumours in adults. Lancet. 2003. 361. 323-331.
14. Olson J.D., Riedel E., DeAngelis L.M. Long-term outcome of low-grade oligodendroglioma and mixed glioma. Neurology. 2000. 54. 1442-1448.
15. Beckmann M.J., Prayson R.A. A clinicopathologic study of 30 cases of oligoastrocytoma including p53 immunohistochemistry. Pathology. 1997. 29. 159-164.
16. Ricci P.E., Dungan D.H. Imaging of low- and intermediate-grade gliomas. Semin. Radiat. Oncol. 2001. 11. 103-112.
17. Shaw E.G., Scheithauer B.W., O’Fallon J.R., Davis D.H. Mixed oligoastrocytomas: a survival and prognostic factor analysis. Neurosurgery. 1994. 34. 577-582.
18. Chaulagain D., Smolanka V.I., Smolanka A.V., Havryliv T.S. The impact of extent of resection in surgical outcome of pilomyxoid astrocytoma: a case study. Ukrainian Neurosurgical Journal. 2021. 27(4). 43-48.
19. Klein M., Heimans J.J., Aaronson N.K., van der Ploeg H.H.M., Grit J., Muller M., Postma T.J., Mooij J.J., Boerman R.H., Beute G.N., Ossenkoppele G.J., van Imhoff G.W., Dekker A.W., Jolles J., Slotman B.J., Struikmans H., Taphoorn M.J. Effect of radiotherapy and other treatment related factors on mid-term to long-term cognitive sequelae in low-grade gliomas: a comparative study. Lancet. 2002. 360(9343). 1361-1368.
20. Chaulagain D., Smolanka V., Smolanka A. Intracranial tumors: overview, histological types, symptoms and treatment plans. Int. J. Health Sci. Res. 2021. 11(10). 133-144.
21. Brada M., Viviers L., Abson C., Hines F., Britton J., Ashley S., Sardell S., Traish D., Gonsalves A., Wilkins P., Westbury C. Phase II study of primary temozolomide chemotherapy in patients with WHO grade II gliomas. Ann. Oncol. 2003. 14. 1715-1721.
22. Buckner J.C., Gesme D. Jr, O’Fallon J.R., Hammack J.E., Stafford S., Brown P.D., Hawkins R., Scheithauer B.W., Erickson B.J., Levitt R., Shaw E.G., Jenkins R. Phase II trial of procarbazine, lomustine, and vincristine as initial therapy for patients with low-grade oligodendroglioma or oligoastrocytoma: efficacy and associations with chromosomal abnormalities. J. Clin. Oncol. 2003. 21. 251-255.
23. Mason W.P., Krol G.S., DeAngelis L.M. Low-grade oligodendroglioma responds to chemotherapy. Neurology. 1996. 46. 203-207.
24. Soffietti R., Ruda R., Bradac G.B., Schiffer D. PCV chemotherapy for recurrent oligodendrogliomas and oligoastrocytomas. Neurosurgery. 1998. 43. 1066-1073.
25. Pace A., Vidiri A., Galie E., Carosi M., Telera S., Cianciulli A.M., Canalini P., Giannarelli D., Jandolo B., Carapella C.M. Temozolomide chemotherapy for progressive low-grade glioma: clinical benefits and radiological response. Ann. Oncol. 2003. 14. 1722-1726.
26. Wessels P.H., Weber W.E., Raven G., Ramaekers F.C., Hopman A.H., Twijnstra A. Supratentorial grade II astrocytoma: biological features and clinical course. Lancet Neurol. 2003. 2. 395-403.

/24.jpg)
/25.jpg)